Paper 1 H - SAMPLE SET 1 Q2
2) An atom of aluminium has the symbol
2.1) Give the number of protons, neutrons and electrons in this atom of aluminium. [3 marks]
Number of protons _____________________
Number of neutrons _____________________
Number of electrons _____________________
2.2) Why is aluminium positioned in Group 3 of the periodic table? [1 mark]
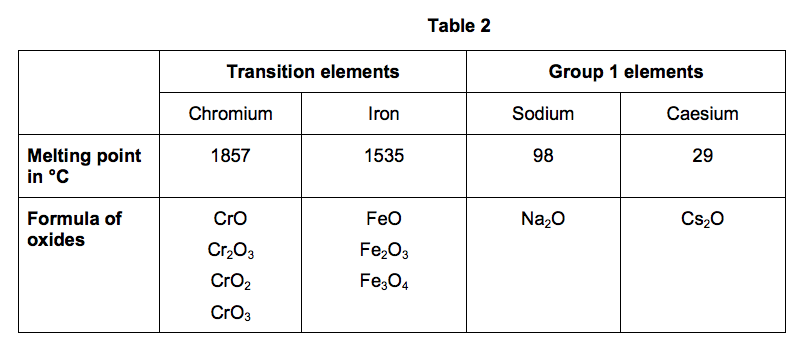
2.3) In the periodic table, the transition elements and Group 1 elements are metals.
Some of the properties of two transition elements and two Group 1 elements are shown in Table 2.
Number of protons _____________________
Number of neutrons _____________________
Number of electrons _____________________
2.2) Why is aluminium positioned in Group 3 of the periodic table? [1 mark]
2.3) In the periodic table, the transition elements and Group 1 elements are metals.
Some of the properties of two transition elements and two Group 1 elements are shown in Table 2.
Use your own knowledge and the data in Table 2 to compare the chemical and physical properties of transition elements and Group 1 elements. [6 marks]
(Total for Question 2 = 10 marks)
(Total for Question 2 = 10 marks)