Paper 1 H - SAMPLE SET 1 Q3
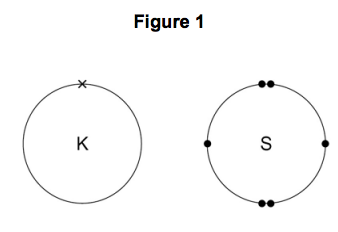
3) Figure 1 shows the outer electrons in an atom of the Group 1 element potassium and in an atom of the Group 6 element sulfur.
3.1) Potassium forms an ionic compound with sulfur.
Describe what happens when two atoms of potassium react with one atom of sulfur.
Give your answer in terms of electron transfer.
Give the formulae of the ions formed. [5 marks]
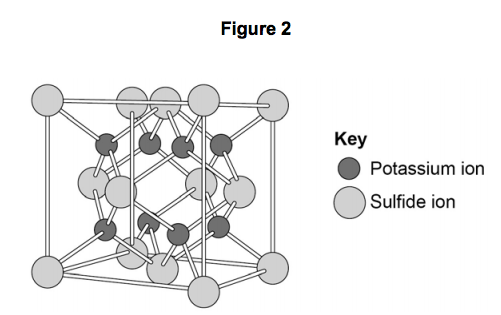
3.2) The structure of potassium sulfide can be represented using the ball and stick model in Figure 2.
Describe what happens when two atoms of potassium react with one atom of sulfur.
Give your answer in terms of electron transfer.
Give the formulae of the ions formed. [5 marks]
3.2) The structure of potassium sulfide can be represented using the ball and stick model in Figure 2.
The ball and stick model is not a true representation of the structure of potassium sulfide.
Give one reason why. [1 mark]
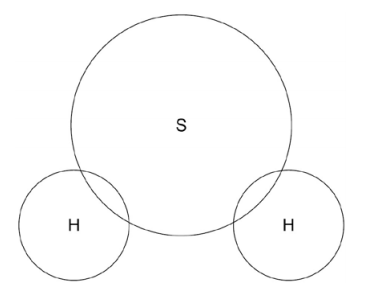
3.3) Sulfur can also form covalent bonds.
Complete the dot and cross diagram to show the covalent bonding in a molecule of hydrogen sulfide.
Show the outer shell electrons only. [2 marks]
Give one reason why. [1 mark]
3.3) Sulfur can also form covalent bonds.
Complete the dot and cross diagram to show the covalent bonding in a molecule of hydrogen sulfide.
Show the outer shell electrons only. [2 marks]
3.4) Calculate the relative formula mass (Mr) of aluminium sulfate, Al2(SO4)3
Relative atomic masses (Ar): oxygen = 16; aluminium = 27; sulfur = 32 [2 marks]
Relative atomic masses (Ar): oxygen = 16; aluminium = 27; sulfur = 32 [2 marks]
Relative formula mass = ____________________________
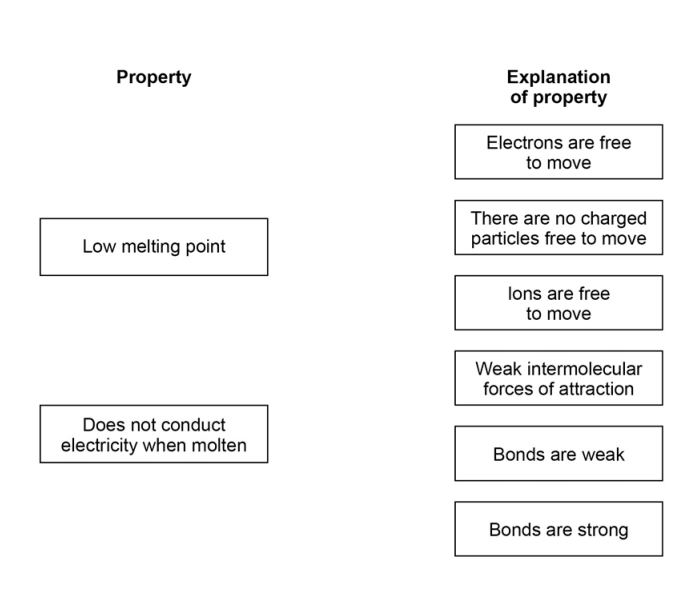
3.5) Covalent compounds such as hydrogen sulfide have low melting points and do not conduct electricity when molten.
Draw one line from each property to the explanation of the property.
[2 marks]
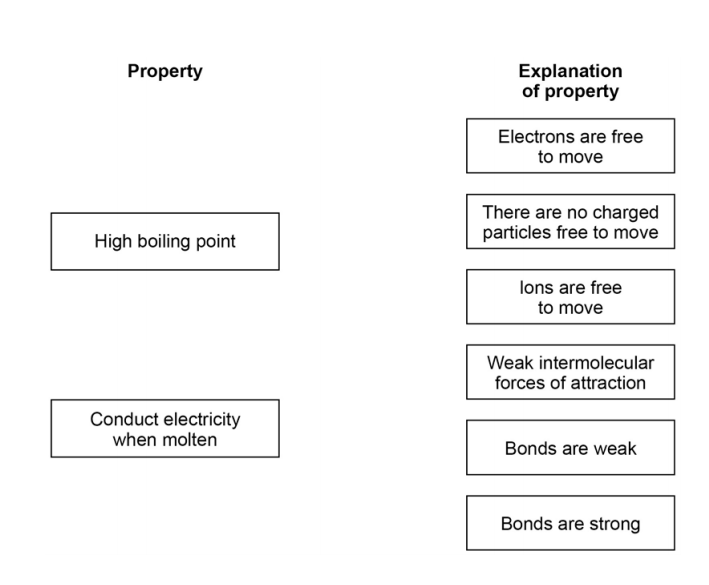
3.6) Ionic compounds such as potassium sulfide have high boiling points and conduct electricity when dissolved in water.
Draw one line from each property to the explanation of the property. [2 marks]
Draw one line from each property to the explanation of the property. [2 marks]
(Total for Question 3 = 14 marks)