Paper 1 H - SAMPLE SET 1 Q4
4) Rock salt is a mixture of sand and salt.
Salt dissolves in water. Sand does not dissolve in water.
Some students separated rock salt.
This is the method used.
1. Place the rock salt in a beaker.
2. Add 100 cm3 of cold water.
3. Allow the sand to settle to the bottom of the beaker.
4. Carefully pour the salty water into an evaporating dish.
5. Heat the contents of the evaporating dish with a Bunsen burner until salt crystals start to form.
4.1) Suggest one improvement to step 2 to make sure all the salt is dissolved in the water. [1 mark]
4.2) The salty water in step 4 still contained very small grains of sand.
Suggest one improvement to step 4 to remove all the sand. [1 mark]
4.3) Suggest one safety precaution the students should take in step 5. [1 mark]
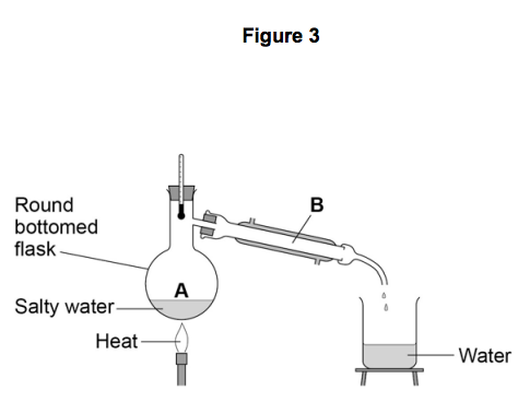
Another student removed water from salty water using the apparatus in Figure 3.
Salt dissolves in water. Sand does not dissolve in water.
Some students separated rock salt.
This is the method used.
1. Place the rock salt in a beaker.
2. Add 100 cm3 of cold water.
3. Allow the sand to settle to the bottom of the beaker.
4. Carefully pour the salty water into an evaporating dish.
5. Heat the contents of the evaporating dish with a Bunsen burner until salt crystals start to form.
4.1) Suggest one improvement to step 2 to make sure all the salt is dissolved in the water. [1 mark]
4.2) The salty water in step 4 still contained very small grains of sand.
Suggest one improvement to step 4 to remove all the sand. [1 mark]
4.3) Suggest one safety precaution the students should take in step 5. [1 mark]
Another student removed water from salty water using the apparatus in Figure 3.
4.4) Describe how this technique works by referring to the processes at A and B.
[2 marks]
4.5) What is the reading on the thermometer during this process? [1 mark]
4.5) What is the reading on the thermometer during this process? [1 mark]
__________________ °C
(Total for Question 4 = 6 marks)