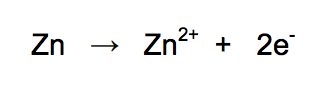Paper 1 H - SAMPLE SET 1 Q6
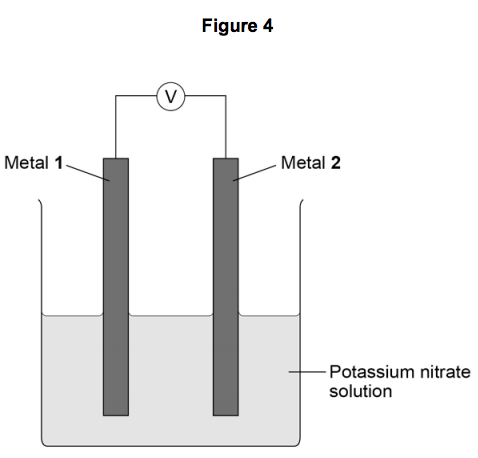
6) A student investigated simple cells using the apparatus shown in Figure 4.
- If metal 2 is more reactive than metal 1 then the voltage measured is positive.
- If metal 1 is more reactive than metal 2 then the voltage measured is negative.
- The bigger the difference in reactivity of the two metals, the larger the voltage produced.
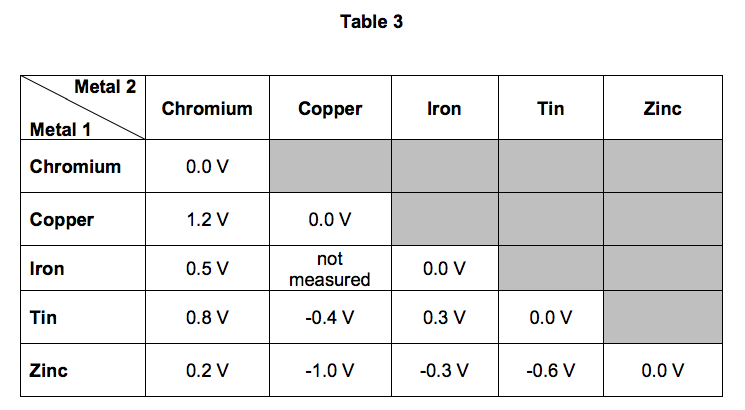
The student’s results are shown in Table 3.
6.1) The ionic equation for the reaction occuring at the zinc electrode in the simple cell made using copper and zinc electrodes is:
Zinc is oxidised in this reaction.
Give a reason why this is oxidation. [1 mark]
6.2) Look at Table 3.
Which one of the metals used was the least reactive?
Give a reason for your answer. [2 marks]
Metal _________________________________
Reason _________________________________
6.3) Predict the voltage that would be obtained for a simple cell that has iron as metal 1 and copper as metal 2.
Explain your answer. [3 marks]
6.4) Hydrogen fuel cells have been developed for cars.
Write a word equation for the overall reaction that takes place in a hydrogen fuel cell. [1 mark]
6.6) Write the two half equations for the reactions that occur at the electrodes in a hydrogen fuel cell. [2 marks]
(Total for Question 6 = 9 marks)
Give a reason why this is oxidation. [1 mark]
6.2) Look at Table 3.
Which one of the metals used was the least reactive?
Give a reason for your answer. [2 marks]
Metal _________________________________
Reason _________________________________
6.3) Predict the voltage that would be obtained for a simple cell that has iron as metal 1 and copper as metal 2.
Explain your answer. [3 marks]
6.4) Hydrogen fuel cells have been developed for cars.
Write a word equation for the overall reaction that takes place in a hydrogen fuel cell. [1 mark]
6.6) Write the two half equations for the reactions that occur at the electrodes in a hydrogen fuel cell. [2 marks]
(Total for Question 6 = 9 marks)