Paper 1 H - SAMPLE SET 1 Q7
Na2CO3 + 2HCl → 2NaCl + H2O + CO2
A student investigated the volume of carbon dioxide produced when different masses of sodium carbonate were reacted with dilute hydrochloric acid.
This is the method used.
1. Place a known mass of sodium carbonate in a conical flask.
2. Measure 10 cm3 of dilute hydrochloric acid using a measuring cylinder.
3. Pour the acid into the conical flask.
4. Place a bung in the flask and collect the gas until the reaction is complete.
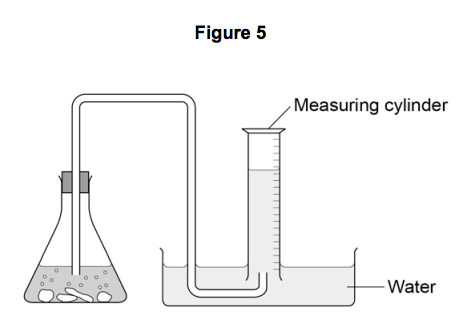
7.1) The student set up the apparatus as shown in Figure 5.
Describe what would happen if the student used the apparatus shown. [2 marks]
The student corrected the error.
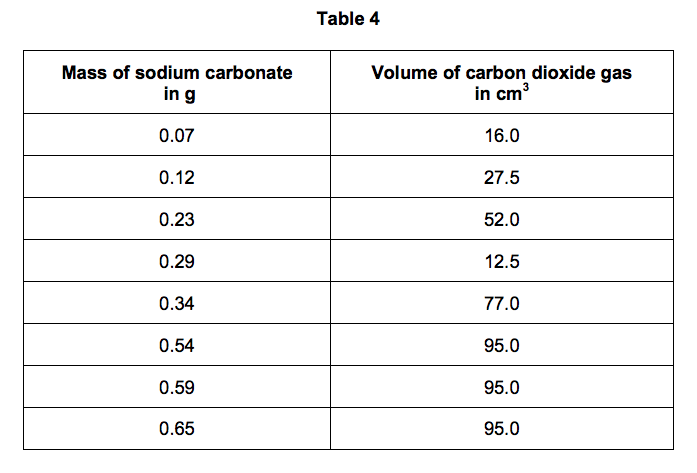
The student’s results are shown in Table 4.
Suggest what may have happened to cause this anomalous result. [1 mark]
7.3) Why does the volume of carbon dioxide collected stop increasing at 95.0 cm3? [1 mark]
7.4) What further work could the student do to be more certain about the minimum mass of sodium carbonate needed to produce 95.0 cm3 of carbon dioxide? [1 mark]
7.5) The carbon dioxide was collected at room temperature and pressure.
The volume of one mole of any gas at room temperature and pressure is 24.0 dm3.
How many moles of carbon dioxide is 95.0 cm3?
Give your answer in three significant figures. [2 marks]
___________________ mol
7.6) Suggest one improvement that could be made to the apparatus used that would give more accurate results.
Give a reason for your answer.
[2 marks]
7.7) One student said that the results of the experiment were wrong because the first few bubbles of gas collected were air.
A second student said this would make no difference to the results.
Explain why the second student was correct.
[2 marks]
(Total for Question 7 = 11 marks)