Paper 1 H - SAMPLE SET 1 Q8
The equation for the reaction is:
2NaOH + H2SO4 → Na2SO4 + 2H2O
8.1) Sulfuric acid is a strong acid.
What is meant by a strong acid?
[2 marks]
8.2) Write the ionic equation for this neutralisation reaction. Include state symbols.
[2 marks]
A student used a pipette to add 25.0 cm3 of sodium hydroxide of unknown concentration to a conical flask.
The student carried out a titration to find out the volume of 0.100 mol/dm3 sulfuric acid needed to neutralise the sodium hydroxide.
8.3) Describe how the student would complete the titration.
You should name a suitable indicator and give the colour change that would be seen.
[4 marks]
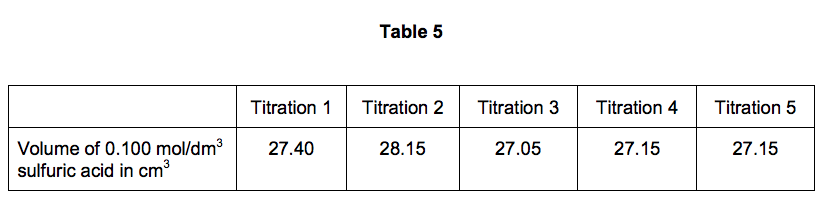
The student carried out five titrations. Her results are shown in Table 5.
Use the student’s concordant results to work out the mean volume of 0.100 mol/dm3 sulfuric acid added. [2 marks]
Mean volume = _________________ cm3
8.5) The equation for the reaction is:
2NaOH + H2SO4 → Na2SO4 + 2H2O
Calculate the concentration of the sodium hydroxide.
Give your answer to three significant figures.
[4 marks]
Concentration = _________________ mol/dm3
8.6) The student did another experiment using 20 cm3 of sodium hydroxide solution with a concentration of 0.18 mol/dm3.
Relative formula mass (Mr) of NaOH = 40
Calculate the mass of sodium hydroxide in 20 cm3 of this solution.
[2 marks]
Mass = _____________ g
(Total for Question 8 = 16 marks)