Paper 1 H - SAMPLE SET 1 Q9
9) This question is about the reaction of ethene and bromine.
The equation for the reaction is:
The equation for the reaction is:
C2H4 + Br2 → C2H4Br2
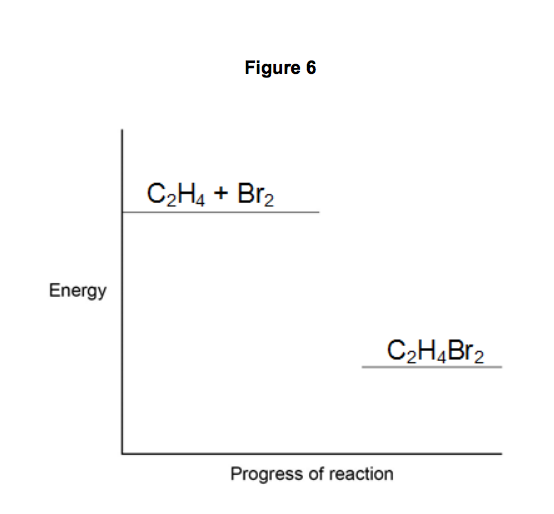
9.1) Complete the reaction profile in Figure 6.
Draw labelled arrows to show:
• The energy given out (∆H)
• The activation energy.
[3 marks]
9.2) When ethene reacts with bromine, energy is required to break covalent bonds in the molecules.
Explain how a covalent bond holds two atoms together. [2 marks]
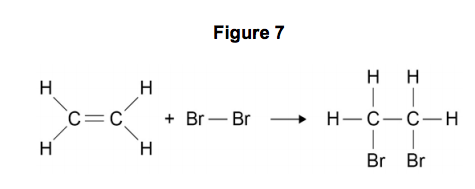
Figure 7 shows the displayed formulae for the reaction of ethene with bromine.
Explain how a covalent bond holds two atoms together. [2 marks]
Figure 7 shows the displayed formulae for the reaction of ethene with bromine.
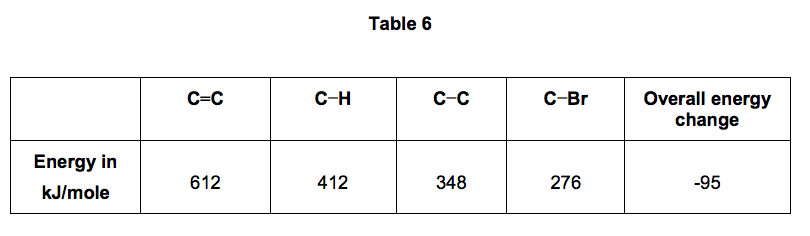
The bond enthalpies and the overall energy change are shown in Table 6.
9.3) Use the information in Table 6 and Figure 7 to calculate the bond energy for the Br−Br bond.
[3 marks]
Bond energy ____________________ kJ/mole
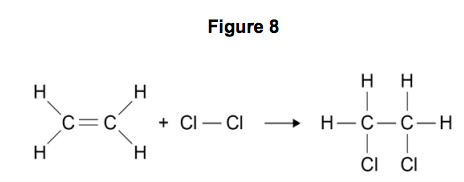
9.4) Figure 8 shows the reaction between ethene and chlorine and is similar to the reaction between ethene and bromine.
“The more energy levels (shells) of electrons an atom has, the weaker the covalent
bonds that it forms.”
Use the above statement to predict and explain how the overall energy change for the reaction of ethene with chlorine will differ from the overall energy change for the reaction of ethene with bromine. [6 marks]
(Total for Question 9 = 14 marks)
Use the above statement to predict and explain how the overall energy change for the reaction of ethene with chlorine will differ from the overall energy change for the reaction of ethene with bromine. [6 marks]
(Total for Question 9 = 14 marks)