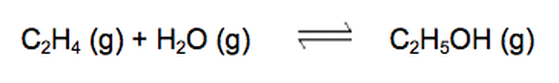Paper 2 H - SAMPLE SET 1 Q10
10) In industry ethanol is produced by the reaction of ethene and steam at 300°C and 60 atmospheres pressure using a catalyst.
The equation for the reaction is:
The equation for the reaction is:
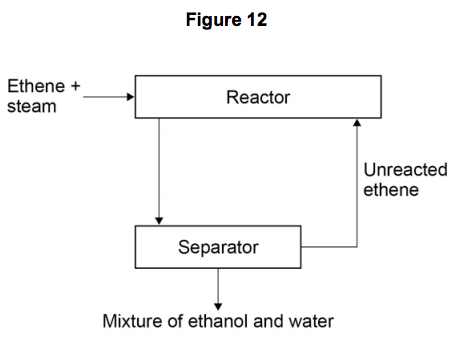
Figure 12 shows a flow diagram of the process.
10.1) Why does the mixture from the separator contain ethanol and water? [1 mark]
10.2) The forward reaction is exothermic.
Use Le Chatelier’s Principle to predict the effect of increasing temperature on the amount of ethanol produced at equilibrium.
Give a reason for your prediction. [2 marks]
10.3) Explain how increasing the pressure of the reactants will affect the amount of ethanol produced at equilibrium. [2 marks]
(Total for Question 10 = 5 marks)
10.2) The forward reaction is exothermic.
Use Le Chatelier’s Principle to predict the effect of increasing temperature on the amount of ethanol produced at equilibrium.
Give a reason for your prediction. [2 marks]
10.3) Explain how increasing the pressure of the reactants will affect the amount of ethanol produced at equilibrium. [2 marks]
(Total for Question 10 = 5 marks)