Paper 2 H - SAMPLE SET 1 Q3
3) A student investigated the specific heat capacity of metals.
3.1) Describe an experiment the student could do to measure the specific heat capacity of a metal. [6 marks]
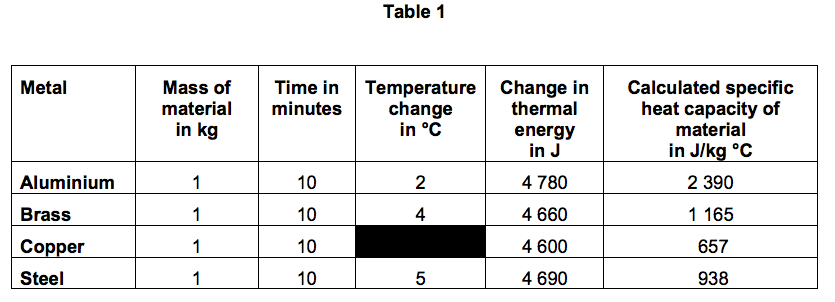
Table 1 shows the student’s results.
3.1) Describe an experiment the student could do to measure the specific heat capacity of a metal. [6 marks]
Table 1 shows the student’s results.
3.2) Use data from Table 1 to calculate the temperature change for copper.
Use the correct equation from the Physics Equation Sheet. [3 marks]
Use the correct equation from the Physics Equation Sheet. [3 marks]
Temperature change = __________ °C
3.3) What is the independent variable in the student’s investigation? [1 mark]
Tick one box.
Tick one box.
3.4) The student calculated the specific heat capacity of aluminium to be 2 390 J/kg °C.
The ‘true’ specific heat capacity of aluminium is 900 J/kg °C.
Suggest why the student’s result for aluminium is different from the ‘true’ value. [2 marks]
3.5) The teacher suggested that putting bubble wrap round the metal block would change the results.
How would using bubble wrap change the results?
Give a reason for your answer. [2 marks]
(Total for Question 3 = 14 marks)
The ‘true’ specific heat capacity of aluminium is 900 J/kg °C.
Suggest why the student’s result for aluminium is different from the ‘true’ value. [2 marks]
3.5) The teacher suggested that putting bubble wrap round the metal block would change the results.
How would using bubble wrap change the results?
Give a reason for your answer. [2 marks]
(Total for Question 3 = 14 marks)