Paper 4 H - SAMPLE SET 1 Q1 Answers
1) A student investigates a potassium salt, X.
She finds that salt X:
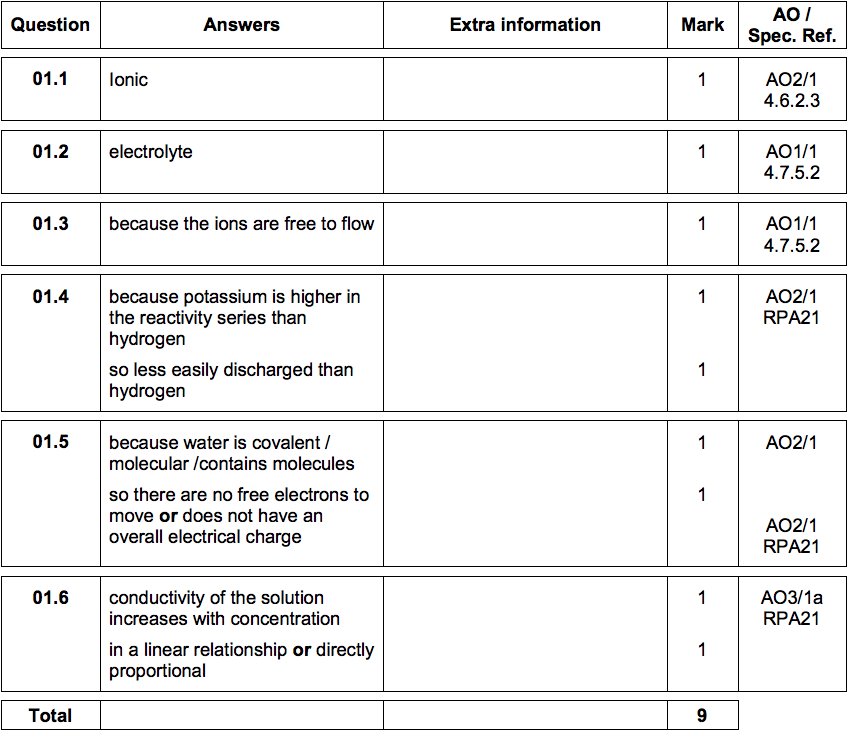
1.1) What is the type of bonding in salt X? [1 mark]
She finds that salt X:
- has a high melting point
- does not conduct electricity when it is solid
- dissolves in water and the solution does conduct electricity.
1.1) What is the type of bonding in salt X? [1 mark]
1.2) What is the name given to solutions that conduct electricity? [1 mark]
1.3) Why does a solution of salt X in water conduct electricity? [1 mark]
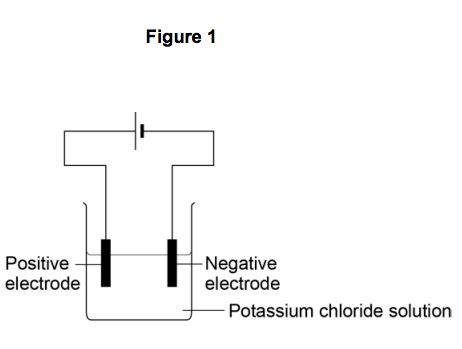
1.4) The student electrolyses a solution of potassium chloride.
Figure 1 shows the apparatus she uses.
1.3) Why does a solution of salt X in water conduct electricity? [1 mark]
1.4) The student electrolyses a solution of potassium chloride.
Figure 1 shows the apparatus she uses.
When the current is switched on, bubbles of hydrogen gas are given off at the negative electrode.
Explain why hydrogen is produced and not potassium. [2 marks]
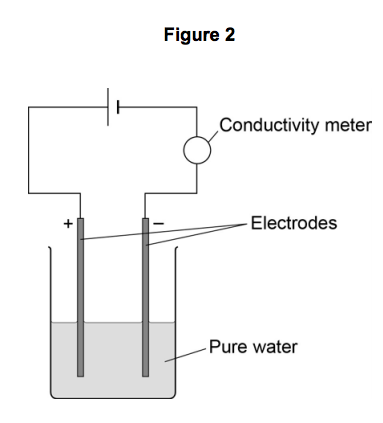
The student then compares the relative conductivity of different concentrations of potassium chloride.
Figure 2 shows the apparatus she uses.
Explain why hydrogen is produced and not potassium. [2 marks]
The student then compares the relative conductivity of different concentrations of potassium chloride.
Figure 2 shows the apparatus she uses.
This is the method used.
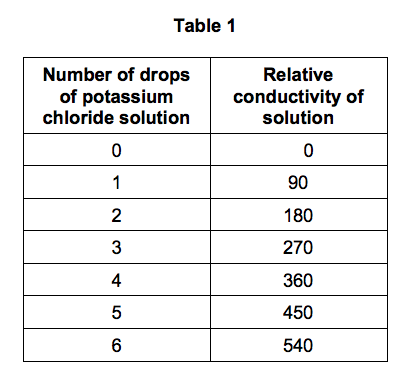
Table 1 shows the student’s results.
- Add potassium chloride solution to the water one drop at a time.
- Stir the mixture.
- Record the reading on the conductivity meter.
Table 1 shows the student’s results.
1.5) When there is no potassium chloride in the beaker no electrical charge flows.
Suggest why pure water does not conduct electricity. [2 marks]
1.6) Describe the relationship shown in Table 1. [2 marks]
(Total for Question 1 = 9 marks)
Suggest why pure water does not conduct electricity. [2 marks]
1.6) Describe the relationship shown in Table 1. [2 marks]
(Total for Question 1 = 9 marks)