Paper 4 H - SAMPLE SET 1 Q5 Answers
5) The elements in Group 1 of the periodic table are metals.
5.1) The elements in Group 1 are called the alkali metals.
Why are they called the alkali metals? [2 marks]
5.2) Explain the increase in reactivity of elements further down the group. [4 marks]
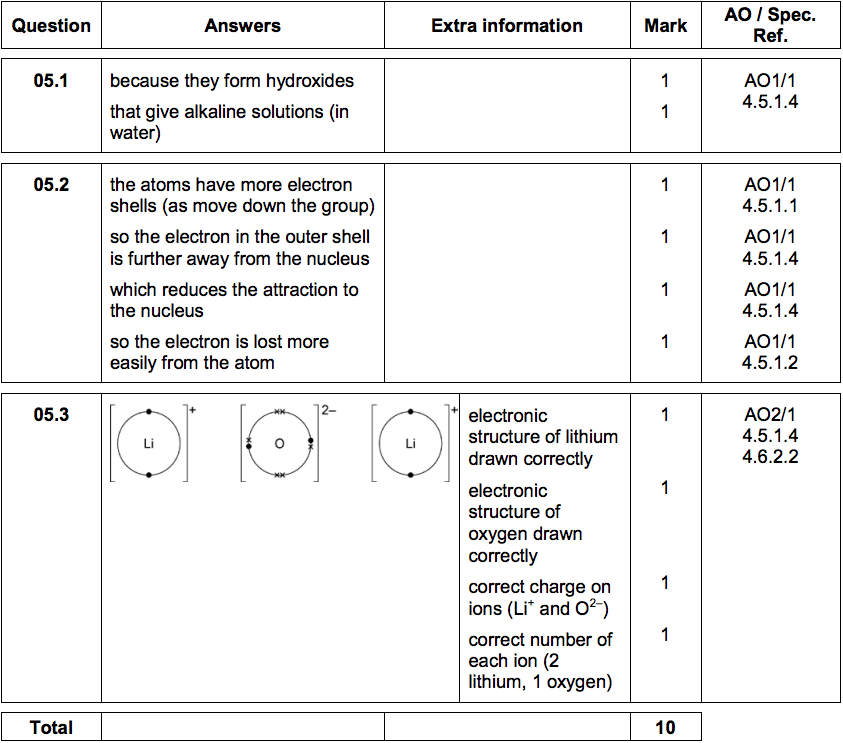
5.3) Lithium oxide is an ionic compound.
Draw a dot and cross diagram to show how lithium and oxygen combine to form lithium oxide.
Only show the electrons in the outer shell of each atom.
Give the charges on the ions formed. [4 marks]
(Total for Question 5 = 10 marks)
5.1) The elements in Group 1 are called the alkali metals.
Why are they called the alkali metals? [2 marks]
5.2) Explain the increase in reactivity of elements further down the group. [4 marks]
5.3) Lithium oxide is an ionic compound.
Draw a dot and cross diagram to show how lithium and oxygen combine to form lithium oxide.
Only show the electrons in the outer shell of each atom.
Give the charges on the ions formed. [4 marks]
(Total for Question 5 = 10 marks)