Paper 4 H - SAMPLE SET 1 Q6
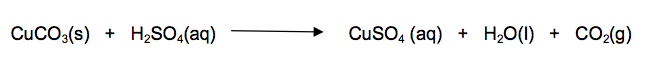
6) The salt copper sulfate can be made by reacting copper carbonate with dilute sulfuric acid.
6.1) Write a method that a student could use to prepare a pure, dry sample of copper
You do not need to write a risk assessment or include safety points. [6 marks]
6.2) Calculate the number of molecules in 14 g of carbon dioxide.
Give your answer in standard form.
Relative atomic masses (Ar): C = 14; O = 16 [4 marks]
You do not need to write a risk assessment or include safety points. [6 marks]
6.2) Calculate the number of molecules in 14 g of carbon dioxide.
Give your answer in standard form.
Relative atomic masses (Ar): C = 14; O = 16 [4 marks]
Answer = _________________ molecules
(Total for Question 6 = 10 marks)