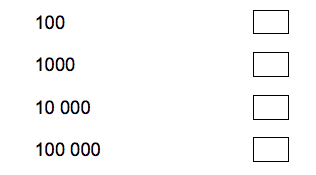4.2 - SPECIMEN SET P1 Q4
4) Atoms are very small and most of their mass is concentrated in the nucleus.
Electrons orbit at different distances from the nucleus.
4.1) A nucleus is much smaller than an atom. Approximately how many times smaller is a nucleus than an atom? [1 mark]
Tick one box.
Electrons orbit at different distances from the nucleus.
4.1) A nucleus is much smaller than an atom. Approximately how many times smaller is a nucleus than an atom? [1 mark]
Tick one box.
4.2) The electrons in an atom can only orbit at specific distances from the nucleus.
State what causes an electron’s distance from the nucleus to increase or decrease. [2 marks]
Increase ________________________________________
Decrease ________________________________________
4.3) Atoms have different atomic numbers and mass numbers.
In terms of sub-atomic particles, describe the difference between an atom’s atomic number and its mass number. [2 marks]
4.4) Transmutation is the name given to a process where one element changes into another.
Explain and compare how two different types of radioactive decay can cause transmutation. [4 marks]
(Total for Question 4 = 9 marks)
State what causes an electron’s distance from the nucleus to increase or decrease. [2 marks]
Increase ________________________________________
Decrease ________________________________________
4.3) Atoms have different atomic numbers and mass numbers.
In terms of sub-atomic particles, describe the difference between an atom’s atomic number and its mass number. [2 marks]
4.4) Transmutation is the name given to a process where one element changes into another.
Explain and compare how two different types of radioactive decay can cause transmutation. [4 marks]
(Total for Question 4 = 9 marks)