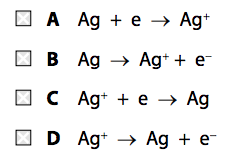C1 H - Sample set 1 Q5
5) Objects made from transition metals are sometimes coated with a thin layer of another transition metal to improve their appearance and to protect against corrosion.
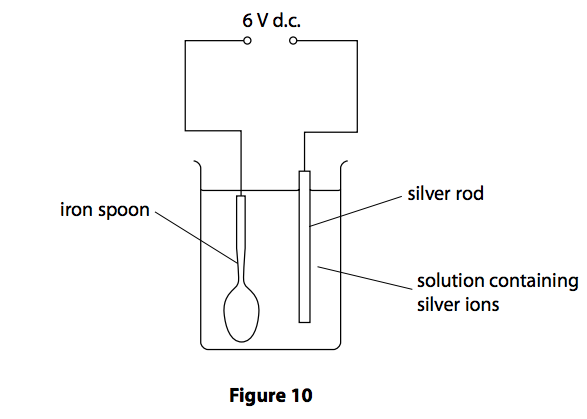
(a) Figure 10 shows equipment that can be used to electroplate an iron spoon with silver.
(a) Figure 10 shows equipment that can be used to electroplate an iron spoon with silver.
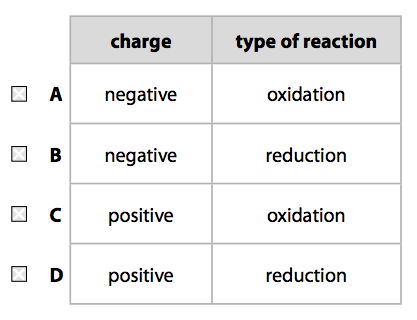
(i) Which row of the table correctly shows the charge on the silver rod electrode and the type of reaction occurring at this electrode? (1)
(ii) Silver metal is deposited on the spoon.
Which half-equation represents this reaction? (1)
Which half-equation represents this reaction? (1)
(b) The voltage of a cell is 1.5V.
Give a reason why this voltage of the cell decreases when the cell is left connected in a circuit. (1)
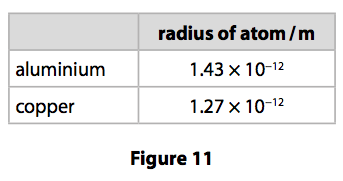
(c) Duralumin is an alloy of aluminium and copper.
The radii of the aluminium and copper atoms are shown in Figure 11.
Give a reason why this voltage of the cell decreases when the cell is left connected in a circuit. (1)
(c) Duralumin is an alloy of aluminium and copper.
The radii of the aluminium and copper atoms are shown in Figure 11.
Explain why copper added to aluminium to form the alloy makes the alloy stronger than pure aluminium. (2)
(d) Gold is often alloyed with other metals when it is used to make jewellery.
The proportion of gold in a piece of gold jewellery is measured in carats.
Pure gold is 24 carats.
A 9 carat gold ring has a mass of 12g.
Calculate the mass of gold in this ring. (2)
(d) Gold is often alloyed with other metals when it is used to make jewellery.
The proportion of gold in a piece of gold jewellery is measured in carats.
Pure gold is 24 carats.
A 9 carat gold ring has a mass of 12g.
Calculate the mass of gold in this ring. (2)
mass of gold ring = ........................................................ g
(Total for Question 5 = 7 marks)