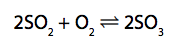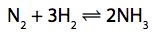C1 H - Sample set 1 Q7
7) Sulfur trioxide is produced by reacting sulfur dioxide with oxygen.
(a) (i) This reaction takes place in industry at 1–2 atm pressure and can reach a
dynamic equilibrium.
Explain the effect on the rate of attainment of equilibrium, if the process is carried out at a pressure higher than 1–2 atm. (3)
(ii) What volume of oxygen, in cm3 , would react completely with 500cm3 sulfur dioxide? (1)
Explain the effect on the rate of attainment of equilibrium, if the process is carried out at a pressure higher than 1–2 atm. (3)
(ii) What volume of oxygen, in cm3 , would react completely with 500cm3 sulfur dioxide? (1)
(b) When there are alternative methods of producing a product, the final pathway is
chosen by considering atom economy, cost of energy, yield of product and rates
of reactions.
State another factor that should also be considered. (1)
*(c)The reaction between nitrogen and hydrogen is exothermic.
State another factor that should also be considered. (1)
*(c)The reaction between nitrogen and hydrogen is exothermic.
If nitrogen and hydrogen were reacted at 150atm pressure and 300°C, without a
catalyst, some ammonia would be formed.
In the Haber process a pressure of 150atm and a temperature of 450°C are used, in the presence of an iron catalyst.
Explain why the conditions used in the Haber process are better than the first set of conditions for the manufacture of ammonia. (6)
(Total for Question 7 = 11 marks)
In the Haber process a pressure of 150atm and a temperature of 450°C are used, in the presence of an iron catalyst.
Explain why the conditions used in the Haber process are better than the first set of conditions for the manufacture of ammonia. (6)
(Total for Question 7 = 11 marks)