C2 H - Sample set 1 Q9
9) (a) A student carried out an experiment to prove that candle wax, a hydrocarbon,
produces carbon dioxide and water vapour when it burns.
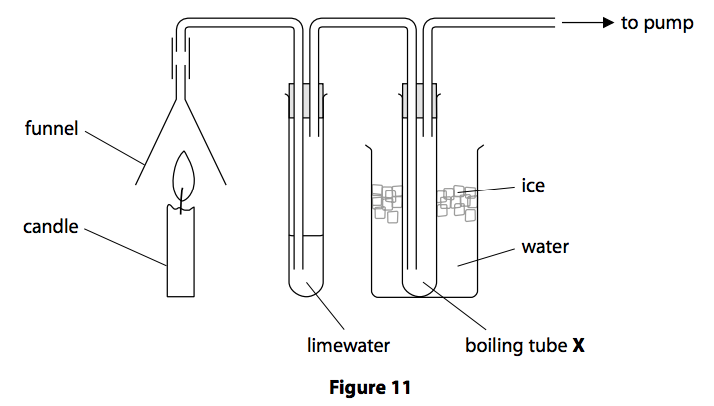
The equipment used is shown in Figure 11.
The gas produced from the burning candle is drawn through the apparatus.
The limewater turned milky showing that carbon dioxide had been formed.
A small amount of a colourless liquid condensed in boiling tube X.
The student claimed that this proved that burning candle wax produced water.
The teacher said the apparatus had been set up incorrectly and therefore this conclusion about water was not valid.
Explain how the student could modify the equipment to prove that water is produced by burning candle wax. (2)
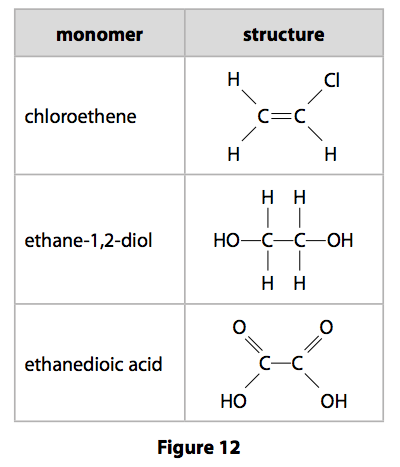
*(b) Polymers are addition or condensation polymers.
Polymers can be formed by using the monomers shown in Figure 12.
A small amount of a colourless liquid condensed in boiling tube X.
The student claimed that this proved that burning candle wax produced water.
The teacher said the apparatus had been set up incorrectly and therefore this conclusion about water was not valid.
Explain how the student could modify the equipment to prove that water is produced by burning candle wax. (2)
*(b) Polymers are addition or condensation polymers.
Polymers can be formed by using the monomers shown in Figure 12.
Explain, using appropriate monomers from Figure 12, how different polymers can
be formed.
(6)
(c) An alcohol A, with molecular formula C2H5OH is oxidised to a compound B with molecular formula C2H4O2.
(i) Compound B is not an alcohol and is a member of another homologous series.
State the name of this homologous series. (1)
(ii) Draw the structure of a molecule of compound A and a molecule of compound B, showing all covalent bonds. (2)
Compound A
Compound B
(Total for Question 9 = 11 marks)
(c) An alcohol A, with molecular formula C2H5OH is oxidised to a compound B with molecular formula C2H4O2.
(i) Compound B is not an alcohol and is a member of another homologous series.
State the name of this homologous series. (1)
(ii) Draw the structure of a molecule of compound A and a molecule of compound B, showing all covalent bonds. (2)
Compound A
Compound B
(Total for Question 9 = 11 marks)