Paper 1 H - SAMPLE SET 1 Q6
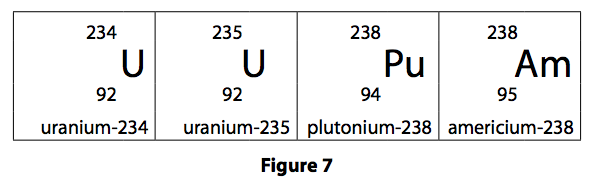
6) Figure 7 shows the nuclei of four atoms.
(a) Which two nuclei have the same number of neutrons? (1)
(b) (i) State what is meant by the term ‘half-life’. (1)
(ii) Plutonium-238 is used in spacecraft to provide heat to power generators.
One of these generators contains 925 g of plutonium-238 when it is manufactured.
One gram of plutonium-238 has a power density of 0.54 W/g.
Plutonium-238 has a half-life of 87.7 years.
Calculate the average energy released per second by the generator after 263 years. (4)
(ii) Plutonium-238 is used in spacecraft to provide heat to power generators.
One of these generators contains 925 g of plutonium-238 when it is manufactured.
One gram of plutonium-238 has a power density of 0.54 W/g.
Plutonium-238 has a half-life of 87.7 years.
Calculate the average energy released per second by the generator after 263 years. (4)
average energy released per second = ............................................... (J)
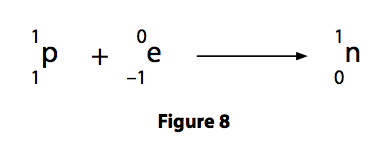
(c) The nucleus of americium-238 can absorb an electron.
When this happens, one of the protons in the nucleus becomes a neutron, as shown in Figure 8.
When this happens, one of the protons in the nucleus becomes a neutron, as shown in Figure 8.
(i) Describe how absorbing an electron affects the proton number and the nucleon number of a nucleus. (2)
(ii) Deduce which nucleus is formed when americium-238 absorbs an electron. (1)
(ii) Deduce which nucleus is formed when americium-238 absorbs an electron. (1)
(Total for Question 6 = 9 marks)