Paper 1 H - SAMPLE SET 1 Q17
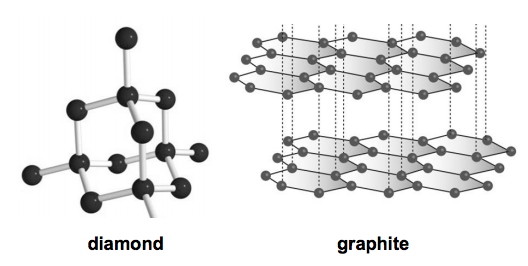
17) (a) The diagrams show the structures of two forms of carbon.
Graphite is a good conductor of electricity.
Diamond does not conduct electricity.
Use ideas about structure and bonding in diamond and graphite to explain these observations. [3]
(b) Carbon can form many thousands of different compounds.
Two examples are shown below.
Diamond does not conduct electricity.
Use ideas about structure and bonding in diamond and graphite to explain these observations. [3]
(b) Carbon can form many thousands of different compounds.
Two examples are shown below.
Why can carbon form many thousands of different compounds? [1]
(c) Ethanol contains carbon.
Look at some information about ethanol.
Melting point = -114°C
Boiling point = 78°C
Predict the state of ethanol at 25°C. How can you tell? [2]
(Total for Question 17 = 6 marks)
(c) Ethanol contains carbon.
Look at some information about ethanol.
Melting point = -114°C
Boiling point = 78°C
Predict the state of ethanol at 25°C. How can you tell? [2]
(Total for Question 17 = 6 marks)