Back to B1 Home
B1 J) Osmosis
B1 J) Osmosis
Substances can move through membranes in 3 different ways; diffusion, osmosis and active transport. We are going to look at osmosis in this section.
Osmosis is the net movement of water molecules through a partially permeable membrane from a region of higher water concentration to a region of lower water concentration.
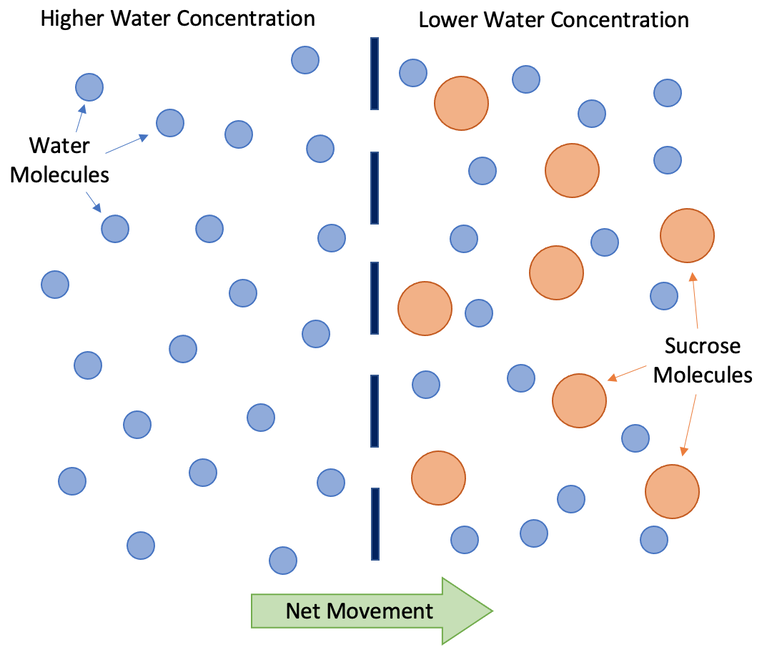
Below is a diagram for osmosis.
The dashed dark blue line down the middle on the above diagram is a partially permeable membrane. The partially permeable membrane has small holes in it. Small substances (like water) can pass through the partially permeable membrane, and larger substances (like sucrose) cannot pass through the partially permeable membrane as they are too large. The water molecules are moving around in random directions, which means that some water molecules will move from left to right and other water molecules will move from right to left.
The overall net movement of water molecules will be from a region of higher water concentration to a region of lower water concentration. On the above diagram, the region of higher water concentration is the left as this is pure water, and the region of lower water concentration is the right as there are sucrose molecules dissolved in the water. This means that the net movement of water molecules will be from left to right.
The overall net movement of water molecules will be from a region of higher water concentration to a region of lower water concentration. On the above diagram, the region of higher water concentration is the left as this is pure water, and the region of lower water concentration is the right as there are sucrose molecules dissolved in the water. This means that the net movement of water molecules will be from left to right.
Why is Osmosis Important?
Osmosis is an important process in the human body as it moves water from cell to cell, and from blood to tissue. It is important that cells in the body are in solutions that have the correct concentrations of solutes. If the concentrations are not correct, the cells can be damaged by osmosis and unable to perform their role/ function.
For example, when red blood cells are placed in pure water, the net movement of water molecules is into the red blood cells through their membranes. This results in the red blood cells swelling up and eventually bursting, thus meaning that the red blood cells cannot carry out their role (which is taking oxygen around the body). On the contrary, when red blood cells are placed in very concentrated salt solutions, the net movement of water molecules is out of the red blood cells through their membranes. This results in the red blood cells shrinking, which also means that the red blood cells cannot carry out their role.
Osmosis is an important process in the human body as it moves water from cell to cell, and from blood to tissue. It is important that cells in the body are in solutions that have the correct concentrations of solutes. If the concentrations are not correct, the cells can be damaged by osmosis and unable to perform their role/ function.
For example, when red blood cells are placed in pure water, the net movement of water molecules is into the red blood cells through their membranes. This results in the red blood cells swelling up and eventually bursting, thus meaning that the red blood cells cannot carry out their role (which is taking oxygen around the body). On the contrary, when red blood cells are placed in very concentrated salt solutions, the net movement of water molecules is out of the red blood cells through their membranes. This results in the red blood cells shrinking, which also means that the red blood cells cannot carry out their role.
Rate of Osmosis
The rate of osmosis is affected by 3 main factors. These factors are essentially the same as the factors that affected the rate of diffusion. The factors are shown below:
The rate of osmosis is affected by 3 main factors. These factors are essentially the same as the factors that affected the rate of diffusion. The factors are shown below:
- Temperature – a higher temperature results in a greater rate of osmosis. This is because a higher temperature gives the water molecules more kinetic energy, which means that they move around faster, thus resulting in a greater rate of osmosis
- Concentration gradient – this refers to the difference in concentrations between the region of high water concentration and the region of low water concentration. A greater concentration gradient for water results in a greater rate of osmosis
- Surface area – the greater the surface area, the greater the rate of osmosis. This is because a greater surface area means that more water molecules can pass through a membrane at once