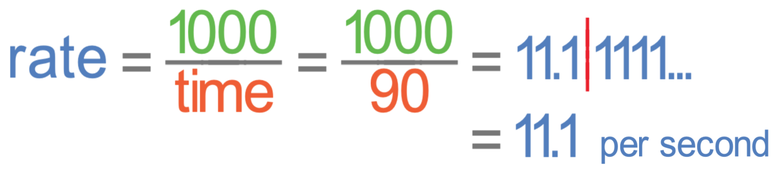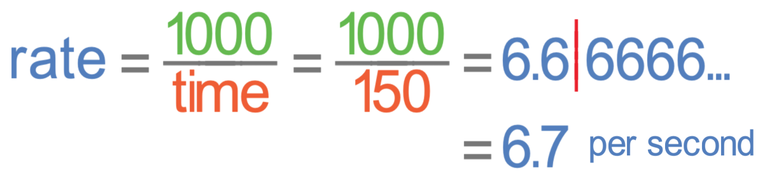Back to B2 Home
B2 D) Enzyme Investigations
B2 D) Enzyme Investigations
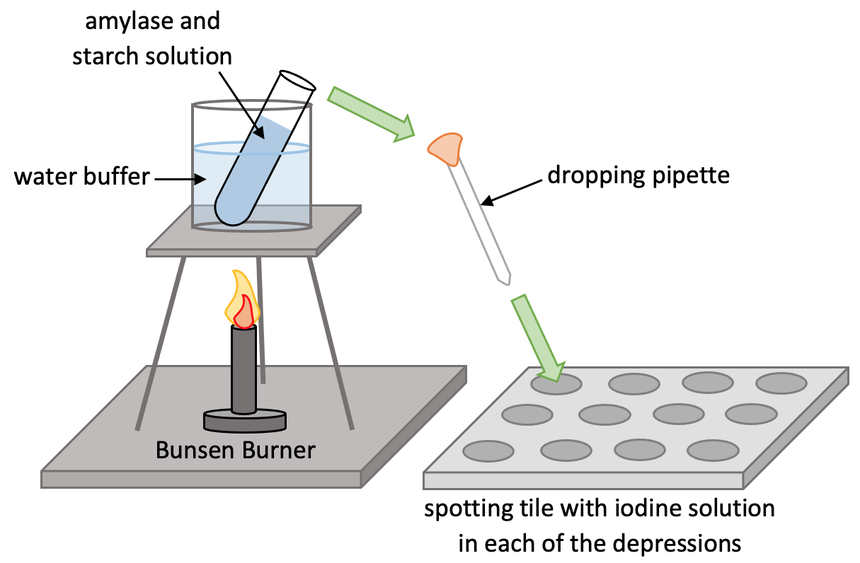
We learnt in the previous section that the rate of reactions for enzymes is affected by temperature and pH. I am going to look at how we can investigate how the rate of reaction for the enzyme amylase is affected by pH. Amylase breaks down starch into the sugar maltose. We will be test different pHs from 5 to 9. The setup for the experiment is shown below.
The experiment uses iodine to test for the presence of starch. Iodine is a browny-orange colour. If starch is present, iodine will change from browny-orange to blue-black. If starch is not present, iodine will stay browny-orange. Before we start the experiment, we fill all of the depressions in the spotting tiles with iodine.
We then fill a beaker with water and place it on top of the tripod. We heat the water to 35°C and we can use a thermometer to check the temperature. We try to keep the temperature of the water at 35°C for the whole experiment. We need to make sure that the temperature of the water does not go too high because the excess temperature will denature the enzymes and affect the whole experiment.
We then fill a beaker with water and place it on top of the tripod. We heat the water to 35°C and we can use a thermometer to check the temperature. We try to keep the temperature of the water at 35°C for the whole experiment. We need to make sure that the temperature of the water does not go too high because the excess temperature will denature the enzymes and affect the whole experiment.
The next step is to place two boiling tubes in the beaker of water for around 5 minutes; one boiling tube will be filled with 5 cm3 of amylase solution and the other will be filled with 5 cm3 of starch suspension. The pH of these solutions will be 5 for the first experiment. These quantities will be the same throughout the whole experiment. We place the two separate test tubes in the beaker of water so that they both get up to 35°C.
After 5 minutes, we pour the amylase solution into the starch suspension creating a mixture of the two. We start a stopwatch as soon as we do this. The amylase will start breaking down the starch in the mixture to produce maltose. After 30 seconds, we use a pipette to transfer a small sample to the depressions that are filled with iodine in the spotting tile. We continue to take samples every 30 seconds for around 5 minutes.
We then note the colour of the depression in the spotting tiles.
- If the depressions in the spotting tile are brown-orange (the same colour as what iodine is), it tells us that no starch present, which means that the amylase enzyme has broken all of the starch down.
- If the depressions in the spotting tile are blue-black, it tells us that starch is still present, which means that the amylase enzyme has not broken all of the starch down yet.
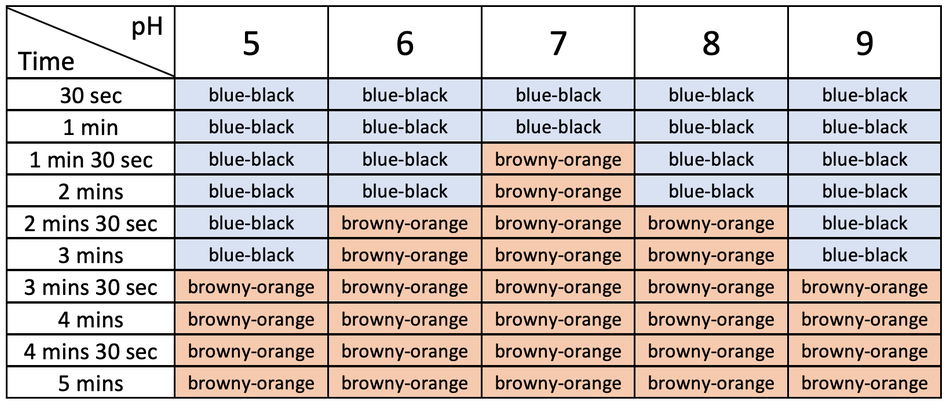
We repeat this whole process for a few different pHs. Our experiment was for a pH of 5, so we repeat it for a pH of 6, 7, 8 and 9. We will then obtain a table of results like what is shown below.
From the table, we can see that for the first few minutes, the colour of all of the spotting tiles were blue-black. This means that starch was present in the sample. We can then see that overtime the colour of the spotting tiles became blue-black. An early time that the spotting tiles stayed browny-orange means that the starch was broken down quicker, thus means that there was a greater rate of reaction for the enzyme. From the table above, we can see that the spotting tile stayed browny-orange after a shorter period of time for a pH of 7. This means that a pH of 7 had the greatest rate of reaction out of all of the different pHs. We can also see that as we go further away from a pH of 7, it took a longer time for the spotting tiles to be browny-orange, thus meaning that pHs further away from 7 had a lower rate of reaction.
Calculating Rate of Reaction
We can calculate the rate of reaction for amylase at different pHs by dividing 1000 by the time taken in seconds. The calculation is shown below.
We can calculate the rate of reaction for amylase at different pHs by dividing 1000 by the time taken in seconds. The calculation is shown below.
pH of 7
I am going to calculate the rate of reaction for a pH of 7. It took 1 minute 30 seconds for amylase to break down all of the starch when amylase was in a pH of 7. The time in the rate of reaction formula needs to be in seconds and this means that we need to convert 1 minute 30 seconds into seconds. There are 60 seconds in 1 minute, which means that 1 minute and 30 seconds is 90 seconds (1 min + 30 sec = 60 sec + 30 sec = 90 sec). We put the time as 90 seconds into the calculation.
I am going to calculate the rate of reaction for a pH of 7. It took 1 minute 30 seconds for amylase to break down all of the starch when amylase was in a pH of 7. The time in the rate of reaction formula needs to be in seconds and this means that we need to convert 1 minute 30 seconds into seconds. There are 60 seconds in 1 minute, which means that 1 minute and 30 seconds is 90 seconds (1 min + 30 sec = 60 sec + 30 sec = 90 sec). We put the time as 90 seconds into the calculation.
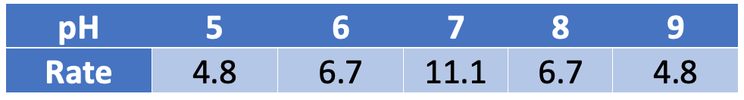
The rate of reaction at a pH of 7 is 11.1 per second.
pH of 8
Let’s now do the same for a pH of 8, which took 2 minutes 30 seconds to break down the starch. 2 minutes and 30 seconds is 150 seconds (2 min + 30 sec = 120 sec + 30 sec = 150 sec). We put the time as 150 seconds into the calculation.
pH of 8
Let’s now do the same for a pH of 8, which took 2 minutes 30 seconds to break down the starch. 2 minutes and 30 seconds is 150 seconds (2 min + 30 sec = 120 sec + 30 sec = 150 sec). We put the time as 150 seconds into the calculation.
The rate of reaction at a pH of 8 is 6.7 per second.
All pHs
The table below shows the rate of reaction for all of the pHs.
All pHs
The table below shows the rate of reaction for all of the pHs.
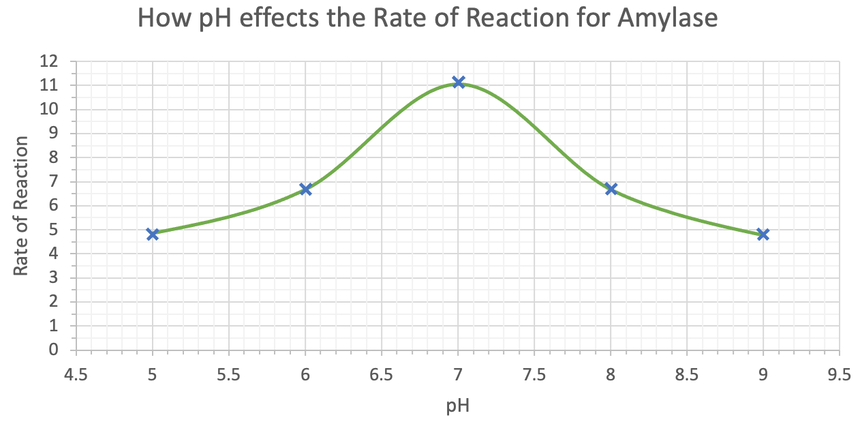
We can now plot these values on a graph. My graph is shown below.
From the above graph, we can see that the optimal pH for amylase is a pH of 7 (neutral). We can tell that this is the optimal pH because amylase has the greatest rate of reaction at this pH. The rate of reaction of amylase decreases as we move away from these pHs, and the further away the pH is from 7, the greater the decrease is in the rate of reaction for amylase. The rate of reaction decreases as we move away from a pH of 7 because the enzymes denature. Denaturing is where the pH causes the bonds in the enzyme to break, which changes the shape of the active site. The substrate (in this case starch) is then unable to fit into the deformed active site, which means that the rate of reaction for the enzyme decreases. Denaturing happens on both sides of the optimal pH; it happens when the pH is less than 7 (6 and 5), and when the pH is greater than 7 (8 and 9).
Investigating Temperature
We can also use this experiment the investigate the affect that temperature has on the rate of reaction for an enzyme. We do this by completing the experiment at a few different temperatures, such as 20°C, 30°C, 40°C, 50°C and 60°C. We then keep the pHs of the starch and amylase solutions at a pH of 7. We also keep the concentrations and volumes of the starch and amylase solutions constant.
We can also use this experiment the investigate the affect that temperature has on the rate of reaction for an enzyme. We do this by completing the experiment at a few different temperatures, such as 20°C, 30°C, 40°C, 50°C and 60°C. We then keep the pHs of the starch and amylase solutions at a pH of 7. We also keep the concentrations and volumes of the starch and amylase solutions constant.