Back to B3 Home
B3 H) Developing Drugs
B3 H) Developing Drugs
We need to vigorously test drugs for a variety of different reasons. Here are the three main reasons.
The testing of drugs can be split into 3 different stages.
- Toxicity – this refers to how harmful the drug is. Is the drug safe for human consumption? Are there any undesired side effects?
- Efficacy – this refers to whether the drug has the desired effect on the body. Does the drug solve the issue that it is trying to solve?
- Dosage – what is the optimal dosage for the drug? This will be the optimal dosage that produces the desired effect (efficacy) whilst having as few side effects as possible (toxicity).
The testing of drugs can be split into 3 different stages.
Stage 1
We test the drug on human cells and tissue in a lab. We can only do this for drugs that have an effect on one type of tissue; we cannot do this for drugs that affect the whole body or organ systems. For example, you would not be able to test a drug that reduces blood pressure on tissue because you would need the whole circulatory system in order to test the drug.
We test the drug on human cells and tissue in a lab. We can only do this for drugs that have an effect on one type of tissue; we cannot do this for drugs that affect the whole body or organ systems. For example, you would not be able to test a drug that reduces blood pressure on tissue because you would need the whole circulatory system in order to test the drug.
Stage 2
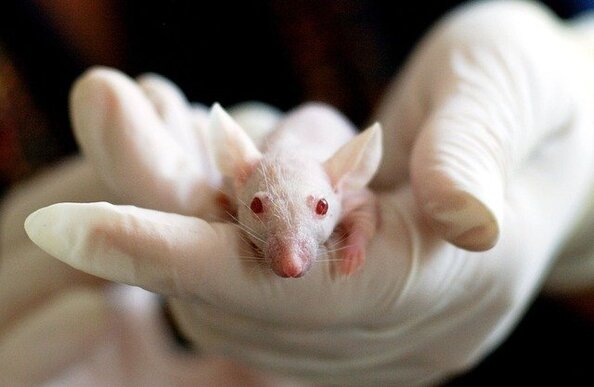
After successful tests on human cells (if we can test the drug on human cells), the drug is then tested on live animals for 3 reasons. These are:
British law requires drugs to be tested on two different types of live mammals before the drug can move onto the next stage of testing (human trials). Some campaigners argue that testing drugs on animals is cruel, but it is the safest way to check that the drug is not dangerous before giving it to humans in the next stage of testing (human trials).
After successful tests on human cells (if we can test the drug on human cells), the drug is then tested on live animals for 3 reasons. These are:
- a) To test efficacy (does the drug produce the effect that you want)
- b) To see if the drug has any toxicity (how harmful is the drug)
- c) To find the optimal dosage (what dosage is most effective)
British law requires drugs to be tested on two different types of live mammals before the drug can move onto the next stage of testing (human trials). Some campaigners argue that testing drugs on animals is cruel, but it is the safest way to check that the drug is not dangerous before giving it to humans in the next stage of testing (human trials).
Stage 3
The next stage of testing is clinical trials.
Healthy Volunteers
First the drug is tested on healthy volunteers to check that the drug is safe. These healthy volunteers are given a very small dosage of the drug and are monitored to see if there are any side effects of the drug. The dosage is slowly increased if the initial data from the drug is positive (no serious side effects).
The drug is then tested on individuals with the illness that the drug is trying to solve. This is to find the optimal dosage of the drug. The optimal dosage is the quantity of the drug that gives the desired effect with few side effects (there are usually some side effects from taking a drug).
Main Clinical Trials
When the optimal dosage has been found, we move onto the main part of testing which is clinical trials. In a clinical trial, the volunteers are split into two groups. One group is given the drug (known as the test group), and the other group is given a placebo (known as the control group). A placebo is a sugar pill that has no effect.
Clinical trials can be blind or double blind. A blind trial is where the volunteers do not know if they are taking the drug or the placebo, but the researchers do know who is taking the drug and who is taking the placebo. Double blind is where both the volunteers and researchers do not know who is taking the drug and who is taking the placebo. Double blind trials exist so that researchers do not give away clues as to which group the volunteers are in (the test group or the control group). If the researchers did give clues, it may change how a volunteer feels and this can make the data less reliable.
The researchers gather data from the test group and the control group. They then compare the data to see how effective the drug is. The presence of the control group who receive the placebo allows for the placebo effect, which is the effect that occurs when a patient feels better because they expect the drug to make them feel better. After the data from the two groups has been compared, the researchers will produce a report that will be peer reviewed to ensure that everything in the clinical trials was carried out correctly and other researchers agree with the claims that the report says; the peer reviewing ensures that the repot is not bias.
The next stage of testing is clinical trials.
Healthy Volunteers
First the drug is tested on healthy volunteers to check that the drug is safe. These healthy volunteers are given a very small dosage of the drug and are monitored to see if there are any side effects of the drug. The dosage is slowly increased if the initial data from the drug is positive (no serious side effects).
The drug is then tested on individuals with the illness that the drug is trying to solve. This is to find the optimal dosage of the drug. The optimal dosage is the quantity of the drug that gives the desired effect with few side effects (there are usually some side effects from taking a drug).
Main Clinical Trials
When the optimal dosage has been found, we move onto the main part of testing which is clinical trials. In a clinical trial, the volunteers are split into two groups. One group is given the drug (known as the test group), and the other group is given a placebo (known as the control group). A placebo is a sugar pill that has no effect.
Clinical trials can be blind or double blind. A blind trial is where the volunteers do not know if they are taking the drug or the placebo, but the researchers do know who is taking the drug and who is taking the placebo. Double blind is where both the volunteers and researchers do not know who is taking the drug and who is taking the placebo. Double blind trials exist so that researchers do not give away clues as to which group the volunteers are in (the test group or the control group). If the researchers did give clues, it may change how a volunteer feels and this can make the data less reliable.
The researchers gather data from the test group and the control group. They then compare the data to see how effective the drug is. The presence of the control group who receive the placebo allows for the placebo effect, which is the effect that occurs when a patient feels better because they expect the drug to make them feel better. After the data from the two groups has been compared, the researchers will produce a report that will be peer reviewed to ensure that everything in the clinical trials was carried out correctly and other researchers agree with the claims that the report says; the peer reviewing ensures that the repot is not bias.
An Example of Testing Going Wrong
Drugs are tested very rigorously to ensure that the drugs are safe before they can be used by doctors/ purchased from shops. However, thalidomide is an example of a drug that was not tested thoroughly enough before it was approved and allowed to be used. Thalidomide was developed in the 1950s to be a sleeping pill. Later it was found to be effective in relieving the effects of morning sickness in pregnant women. However, thalidomide had not been tested to see what effect it had on pregnant women, and therefore it was not known that chemicals from the drug could pass through the placenta to the fetus. The limbs of babies of pregnant women who took thalidomide were affected and some babies were born with no limbs at all. Thalidomide was banned, and it led to laws that require more rigorous testing of drugs before they get approved. More recently, thalidomide has been successfully used to treat leprosy and other diseases.
Drugs are tested very rigorously to ensure that the drugs are safe before they can be used by doctors/ purchased from shops. However, thalidomide is an example of a drug that was not tested thoroughly enough before it was approved and allowed to be used. Thalidomide was developed in the 1950s to be a sleeping pill. Later it was found to be effective in relieving the effects of morning sickness in pregnant women. However, thalidomide had not been tested to see what effect it had on pregnant women, and therefore it was not known that chemicals from the drug could pass through the placenta to the fetus. The limbs of babies of pregnant women who took thalidomide were affected and some babies were born with no limbs at all. Thalidomide was banned, and it led to laws that require more rigorous testing of drugs before they get approved. More recently, thalidomide has been successfully used to treat leprosy and other diseases.