Back to C10 Home
C10 D) Potable Water
C10 D) Potable Water
Potable water is water that is safe for humans to drink. Potable water is not pure water. Pure water only contains H2O molecules and no other substances. Whereas, potable water contains other dissolved substances, such as salts. Potable water can occur naturally, or it can be obtained from other water sources (the water from these other water sources will need to be treated to make the water drinkable/ potable; the treating of water removes harmful substances such as chemicals and pathogens).
Potable water has a pH that is between 6.5 and 8.5. Water with a pH that is outside of this range is harmful for humans to drink.
Potable water has a pH that is between 6.5 and 8.5. Water with a pH that is outside of this range is harmful for humans to drink.
Purifying Water
There are a variety of different water sources that we can treat so that the water is safe to drink. The water source that we use to obtain potable water depends on the availability of the water source and the cost.
Fresh Water
Rainwater is fresh water, which means that it is water that does not have many substances dissolved in it. Rainwater lands on the ground and it can be stored as surface water (rivers, lakes and reservoirs) or end up as ground water (water that is held underground in cracks and spaces in soil and rocks).
The fresh water source that is used to obtain water in the UK depends on the location within the UK. The south of the UK is hotter than the North of the UK. This means that surface water tends to evaporate quicker in the south of the UK compared to the North of the UK. This results in there being less surface water available in the south of the UK compared to the North of the UK. Therefore, potable water in the south of the UK is more likely to come from groundwater rather than surface water (lakes, reservoirs and rivers). Whereas, potable water in the north is more likely to come from surface water rather than ground water.
There are a variety of different water sources that we can treat so that the water is safe to drink. The water source that we use to obtain potable water depends on the availability of the water source and the cost.
Fresh Water
Rainwater is fresh water, which means that it is water that does not have many substances dissolved in it. Rainwater lands on the ground and it can be stored as surface water (rivers, lakes and reservoirs) or end up as ground water (water that is held underground in cracks and spaces in soil and rocks).
The fresh water source that is used to obtain water in the UK depends on the location within the UK. The south of the UK is hotter than the North of the UK. This means that surface water tends to evaporate quicker in the south of the UK compared to the North of the UK. This results in there being less surface water available in the south of the UK compared to the North of the UK. Therefore, potable water in the south of the UK is more likely to come from groundwater rather than surface water (lakes, reservoirs and rivers). Whereas, potable water in the north is more likely to come from surface water rather than ground water.
Treating Water from Surface or Ground Water
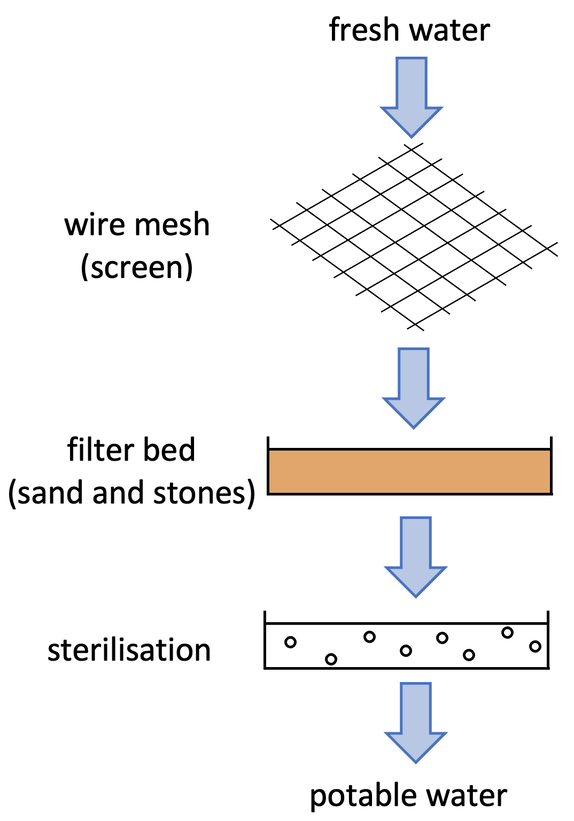
Fresh water from surface water or ground water still needs to be treated despite it being fresh water with low levels of dissolved substances. We treat fresh water to remove potential chemicals and pathogens. The process in treating fresh water involves filtration and sterilisation.
Filtration
The fresh water passes through a wire mesh, which is also known as a screen. The mesh removes large pieces of debris in the water, such as twigs and weeds. The water then passes through a filter bed that contains particles of various sizes from stones to sand. As the water passes through the filter bed, very small particles are removed, and other organic matter is broken down.
Fresh water from surface water or ground water still needs to be treated despite it being fresh water with low levels of dissolved substances. We treat fresh water to remove potential chemicals and pathogens. The process in treating fresh water involves filtration and sterilisation.
Filtration
The fresh water passes through a wire mesh, which is also known as a screen. The mesh removes large pieces of debris in the water, such as twigs and weeds. The water then passes through a filter bed that contains particles of various sizes from stones to sand. As the water passes through the filter bed, very small particles are removed, and other organic matter is broken down.
Sterilisation
After the water has been filtered it is sterilised to kill any harmful microorganisms like pathogens, bacteria or microbes. Usually the sterilisation happens by bubbling chlorine gas through the water – the chlorine gas will kill any harmful microorganisms. The sterilisation may also be done with ozone or ultraviolet light. The water is now safe to drink – the water is potable.
Fresh water is the cheapest source of potable water. However, some hot countries do not have much or any fresh water sources and therefore have to obtain potable water from other sources, such as sea water. These other sources of water are considerably more expensive than obtaining potable water from fresh water.
After the water has been filtered it is sterilised to kill any harmful microorganisms like pathogens, bacteria or microbes. Usually the sterilisation happens by bubbling chlorine gas through the water – the chlorine gas will kill any harmful microorganisms. The sterilisation may also be done with ozone or ultraviolet light. The water is now safe to drink – the water is potable.
Fresh water is the cheapest source of potable water. However, some hot countries do not have much or any fresh water sources and therefore have to obtain potable water from other sources, such as sea water. These other sources of water are considerably more expensive than obtaining potable water from fresh water.
Water from Sea Water – Desalination
We can obtain potable water from sea water through a process known as desalination. Desalination is a very expensive process because it uses lots of energy. Therefore, a country would only obtain potable water from sea water if they had very little fresh water available to them. A country that obtains potable water from sea water is the United Arab Emirates – they obtain 99% of their potable water from sea water. We can obtain potable water from sea water by using either distillation or reverse osmosis.
We can obtain potable water from sea water through a process known as desalination. Desalination is a very expensive process because it uses lots of energy. Therefore, a country would only obtain potable water from sea water if they had very little fresh water available to them. A country that obtains potable water from sea water is the United Arab Emirates – they obtain 99% of their potable water from sea water. We can obtain potable water from sea water by using either distillation or reverse osmosis.
Distillation
Distillation involves heating sea water until the water boils. When the water boils, it turns into a gas and leaves the sea water. The water vapour then passes through a condenser where it cools and turns from water vapour back into liquid water. The condensed water vapour is potable water, which can be drunk. Vast quantities of energy are required to both boil the sea water and cool the water vapour. This makes obtaining potable water from sea water very expensive. Also, the energy requirements may have some adverse effects on the environment if the energy is obtained from sources that produce greenhouse gases or other emissions. Another negative of obtaining potable water from sea water by distillation is that the waste water is very salty and is difficult to dispose of; if the waste water was disposed of in the sea, the waste water could affect marine life.
Experiment
We can undertake an experiment to obtain potable water from sea/salty water in school. The first step is to test the pH of the sea water. We test the pH of the sea water because potable water must have a pH between 6.5 and 8.5. We then take the appropriate action to bring the pH within this range/ to get the pH as 7 (neutral). If the sea water is too acidic, we add some alkali. If the sea water is an alkaline above 8.5, we add some acid.
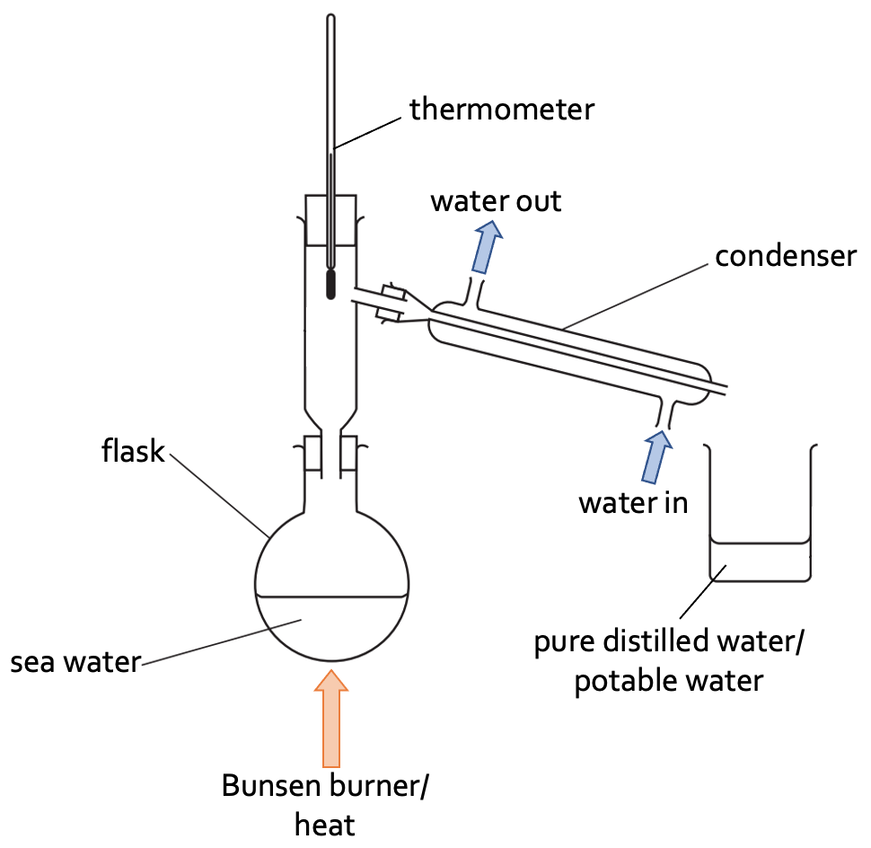
We are now ready to set up the experiment. We place the sea water in a distillation flask and place the flask on top of a tripod with a Bunsen burner underneath. We also attach a rubber pipe from a cold tap to the condenser; the rubber tap goes into the bottom of the condenser and comes out at the top of the condenser. The apparatus is set up like what is shown below.
Distillation involves heating sea water until the water boils. When the water boils, it turns into a gas and leaves the sea water. The water vapour then passes through a condenser where it cools and turns from water vapour back into liquid water. The condensed water vapour is potable water, which can be drunk. Vast quantities of energy are required to both boil the sea water and cool the water vapour. This makes obtaining potable water from sea water very expensive. Also, the energy requirements may have some adverse effects on the environment if the energy is obtained from sources that produce greenhouse gases or other emissions. Another negative of obtaining potable water from sea water by distillation is that the waste water is very salty and is difficult to dispose of; if the waste water was disposed of in the sea, the waste water could affect marine life.
Experiment
We can undertake an experiment to obtain potable water from sea/salty water in school. The first step is to test the pH of the sea water. We test the pH of the sea water because potable water must have a pH between 6.5 and 8.5. We then take the appropriate action to bring the pH within this range/ to get the pH as 7 (neutral). If the sea water is too acidic, we add some alkali. If the sea water is an alkaline above 8.5, we add some acid.
We are now ready to set up the experiment. We place the sea water in a distillation flask and place the flask on top of a tripod with a Bunsen burner underneath. We also attach a rubber pipe from a cold tap to the condenser; the rubber tap goes into the bottom of the condenser and comes out at the top of the condenser. The apparatus is set up like what is shown below.
The next step is to turn the Bunsen burner on, which will heat the distillation flask. As the distillation flask is heated up, the water in the sea water evaporates and turns into a gas (water vapour). The water vapour then rises up the distillation flask and moves down the condenser. The cold water on the outside of the condenser keeps the condenser cool, which results in the water vapour condensing; the water turns from a gas to a liquid. The water then travels down the condenser and into a beaker.
We then test the pH of the solution in the beaker to check that it is neutral – it should be neutral, but it is always a good idea to check again. If the solution is neutral, we have obtained potable water.
The salts and any other impurities in the sea water will be left at the bottom of the distillation flask – at the end of the experiment, we will see some crystals at the bottom of the distillation flask.
Reverse Osmosis
We can also obtain potable water from sea water by using a process known as reverse osmosis. Reverse osmosis works by firing water at a very high pressure at a membrane. The membrane is partially permeable, which means that the membrane will allow the small water molecules through but will not allow the much larger ions and other molecules to pass through. The water that passes through the membrane will be potable water.
Reverse osmosis is a very expensive process for 2 reasons. The first reason is that getting water to a high pressure requires lots of energy. The second is that the membranes involved in reverse osmosis are quite expensive. Like distillation, reverse osmosis produces very salt waste water, which is quite tricky to dispose of.
We then test the pH of the solution in the beaker to check that it is neutral – it should be neutral, but it is always a good idea to check again. If the solution is neutral, we have obtained potable water.
The salts and any other impurities in the sea water will be left at the bottom of the distillation flask – at the end of the experiment, we will see some crystals at the bottom of the distillation flask.
Reverse Osmosis
We can also obtain potable water from sea water by using a process known as reverse osmosis. Reverse osmosis works by firing water at a very high pressure at a membrane. The membrane is partially permeable, which means that the membrane will allow the small water molecules through but will not allow the much larger ions and other molecules to pass through. The water that passes through the membrane will be potable water.
Reverse osmosis is a very expensive process for 2 reasons. The first reason is that getting water to a high pressure requires lots of energy. The second is that the membranes involved in reverse osmosis are quite expensive. Like distillation, reverse osmosis produces very salt waste water, which is quite tricky to dispose of.