C1 A) Atoms
Atoms are tiny particles that make up every item in the world. Atoms are extremely small; they have a radius of about 0.1 nanometres (1 x 10-10 m). Atoms are made out of three subatomic particles:
- Protons
- Neutrons
- Electrons
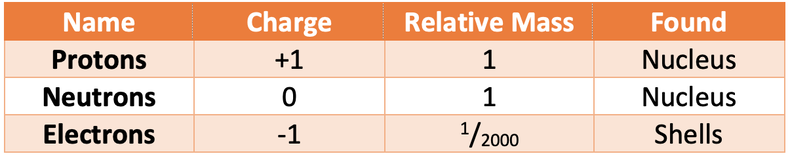
The table below shows the charge, relative mass and location in an atom (found) for protons, neutrons and electrons.
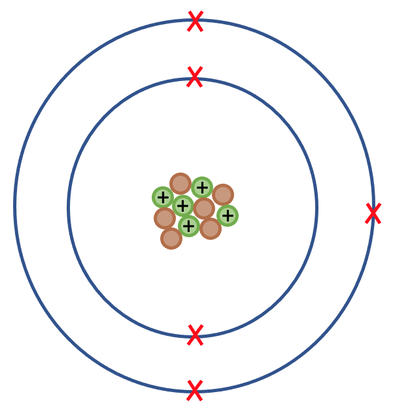
Protons and neutrons are found in the nucleus of an atom (the centre of an atom). The nucleus is tiny in comparison to the size of the rest of the atom; the radius of the nucleus is about 10,000 times smaller than the radius of the atom (the radius of an atom is about 1 x 10-14 m). The nucleus of an atom is positively charged because the protons are found in the nucleus and they have a positive charge (neutrons are also found in the nucleus, but they have no charge and therefore no effect on the charge of the nucleus). Almost all of the mass in an atom is found in the nucleus and this is because all of the protons and neutrons are found in the nucleus.
Electrons move around the nucleus in shells. These shells are referred to as electron shells or energy levels. There are many electron shells and each of the shells can only hold a certain number of electrons. The first shell holds a maximum of 2 electrons, the second and third shells hold a maximum of 8 electrons. An electron will always fill the closest shell to the nucleus. When the closest shell is full, the electrons will start filling the next shell out; for example, if the first shell is full, the electrons will start filling the second shell. Shells do not have to be full. Electrons have virtually no mass; their mass is often taken as zero or 1/2000.
The rest and the majority of the atom is made up of space. Below is a sketch of boron.
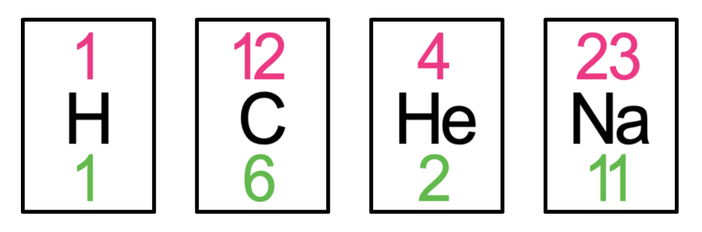
A substance that is made up of only one type of atom is known as an element. There are over 100 different elements. Elements are represented by a one or two letter symbol. This makes it easier and quicker to write out formulas and chemical equations. It is the number of protons that determines what element an atom is. All atoms that have the same number of protons are the same element. For example, all hydrogen atoms have 1 proton and have the symbol H; all carbon atoms have 6 protons and the symbol C; all magnesium atoms have 12 protons and have the symbol Mg.
For all elements, it is only the first letter of the letter symbol that is capitalised; if the element has 2 letters, the second letter will be a lower case. For example, the letter symbol for helium is He; the first letter (H) is a capital and the second letter (e) is lower case. Another example is sodium, which has the symbol Na; the N is capitalised and the a is in lower case.
Elements will have no overall electrical charge. This means that they have the same number of protons and electrons. Protons and electrons have the same magnitude of charge but in opposite directions (protons are positive, and electrons are negative). Atoms that have an unequal number of protons and electrons are charged and we refer to these as ions. If an atom has more protons than electrons, it will be positively charged and referred to as a positive ion. If the atom has fewer protons than electrons, it will be negatively charged and referred to as a negative ion.