Back to C1 Home
C1 B) The Periodic Table
C1 B) The Periodic Table
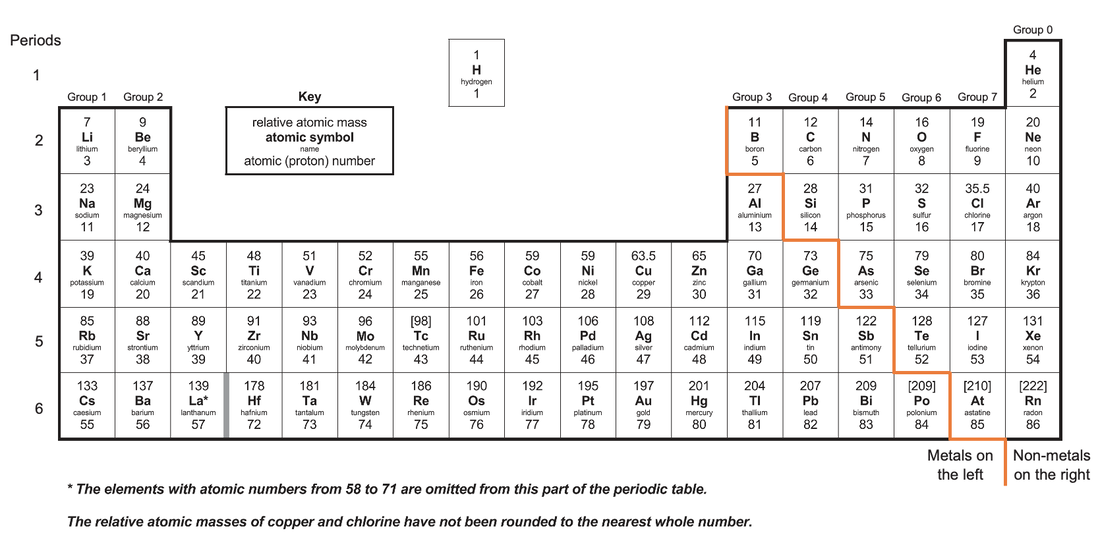
The periodic table shows all elements. The elements are laid out in order of increasing atomic number (the number of protons). The periodic table has a metal and non-metal split; metals are on the left and bottom of the periodic table, and non-metals are on the right and top of the periodic table. The periodic table is laid out in a special way so that the columns and rows tell us certain characteristics about the elements in the periodic table. The rows are referred to as periods and the columns are referred to as groups. The periodic table is shown below.
The periods (rows) tell us how many electron shells the element has.
The groups (columns) tell us how many electrons the elements have in their outermost shell.
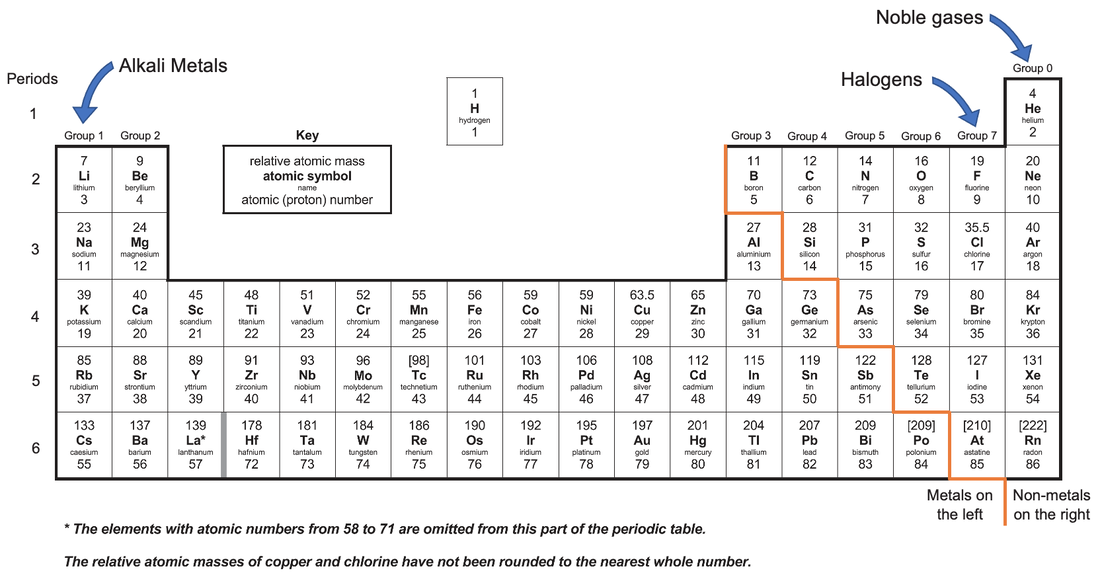
It is useful to know what group an element is in because the group will inform us about how the element will behave/react. For example, all of the group 1 elements (the alkali metals) react very aggressively. All of the group 0 elements (the noble gases) are unreactive. We will be looking at group 1, group 7 (the halogens) and group 0 in more detail later on in this whole subsection.
- Elements in the first period have 1 electron shell; hydrogen and helium are in period 1, so they both have 1 electron shell in use.
- Elements in the second period have 2 electron shells; lithium, beryllium and boron are in period 2, so they will all have 2 electron shells in use.
- Elements in the third period have 3 electron shells; sodium, magnesium and aluminium are all in period 3, so they all have 3 electron shells in use.
The groups (columns) tell us how many electrons the elements have in their outermost shell.
- Elements in group 1 have 1 electron in their outer most shell; lithium, sodium and potassium are all in group 1, so will all have 1 electron in their outermost shell.
- Elements in group 2 have 2 electrons in their outermost shell; beryllium, magnesium and calcium are all in group 2, so will all have 2 electrons in their outermost shell.
- Elements in group 7 have 7 electrons in their outermost shell; fluorine, chlorine and bromine are all in group 7, so will all have 7 electrons in their outermost shell.
- The only exception to this rule is group 0, which tells us that the elements have full outer shells. So, helium, neon and argon all have full outer shells.
It is useful to know what group an element is in because the group will inform us about how the element will behave/react. For example, all of the group 1 elements (the alkali metals) react very aggressively. All of the group 0 elements (the noble gases) are unreactive. We will be looking at group 1, group 7 (the halogens) and group 0 in more detail later on in this whole subsection.