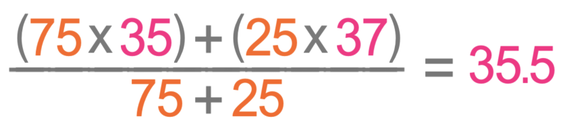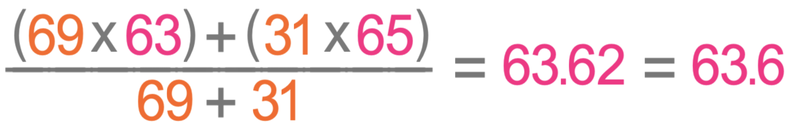Back to C1 Home
C1 D) Isotopes
C1 D) Isotopes
Isotopes are different forms of the same element. As they are the same element, they will have the same number of protons (remember, it is the number of protons that determines what element an atom is). However, the isotopes of an element will have different numbers of neutrons.
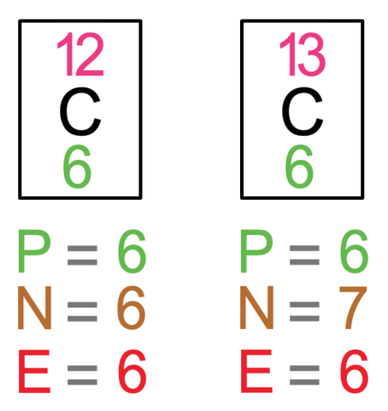
As isotopes have the same number of protons, the atomic numbers will be the same (bottom numbers). And, as isotopes have a different number of neutrons, the mass numbers will be different. A good example of isotopes is carbon-12 and carbon-13. The tiles for these two isotopes and the working for the number of protons (atomic number), neutrons (mass number – atomic number) and electrons (same as the number of protons) are shown below.
As isotopes have the same number of protons, the atomic numbers will be the same (bottom numbers). And, as isotopes have a different number of neutrons, the mass numbers will be different. A good example of isotopes is carbon-12 and carbon-13. The tiles for these two isotopes and the working for the number of protons (atomic number), neutrons (mass number – atomic number) and electrons (same as the number of protons) are shown below.
From comparing the number of protons, neutrons and electrons in carbon-12 and carbon-13, we can see that the number of protons are the same (hence why they are both carbon atoms), the number of electrons are the same, and the number of neutrons are different (carbon-12 has 6 neutrons and carbon-13 has 7 neutrons).
Elements have many different isotopes with different masses and abundances (abundance refers to how common the isotope is; we usually measure abundances in percentages). The relative atomic mass (the mass number; Ar) of an element is an average value for the mass of the element that takes into account the abundances of the various isotopes of the element. We work out the relative atomic for an element by using the formula below.
Elements have many different isotopes with different masses and abundances (abundance refers to how common the isotope is; we usually measure abundances in percentages). The relative atomic mass (the mass number; Ar) of an element is an average value for the mass of the element that takes into account the abundances of the various isotopes of the element. We work out the relative atomic for an element by using the formula below.
Let’s have an example.
Example 1
Chlorine has two isotopes; chlorine-35 has an abundance of 75%, and chlorine-37 has an abundance of 25%. Calculate the relative atomic mass for chlorine giving your answer to one decimal place.
Chlorine-35 has an abundance of 75% and a mass of 35. Chlorine-37 has an abundance of 25% and a mass of 37. We work out the relative atomic mass by placing these values into the formula.
Chlorine has two isotopes; chlorine-35 has an abundance of 75%, and chlorine-37 has an abundance of 25%. Calculate the relative atomic mass for chlorine giving your answer to one decimal place.
Chlorine-35 has an abundance of 75% and a mass of 35. Chlorine-37 has an abundance of 25% and a mass of 37. We work out the relative atomic mass by placing these values into the formula.
The relative atomic mass of chlorine is 35.5.
Example 2
There are two isotopes of copper; copper-63 has an abundance of 69%, and copper-65 has an abundance of 31%. Calculate the relative atomic mass for copper giving your answer to one decimal place.
Copper-63 has an abundance of 69% and a mass of 63. Copper-65 has an abundance of 31% and a mass of 65. We work out the relative atomic mass by placing these values in the formula.
There are two isotopes of copper; copper-63 has an abundance of 69%, and copper-65 has an abundance of 31%. Calculate the relative atomic mass for copper giving your answer to one decimal place.
Copper-63 has an abundance of 69% and a mass of 63. Copper-65 has an abundance of 31% and a mass of 65. We work out the relative atomic mass by placing these values in the formula.
The relative atomic mass of copper is 63.6.