Overtime the model of atoms has changed because new experiments have contradicted the predictions of previous models of atoms. Currently we use what is known as the nuclear model to explain the structure of atoms. The nuclear model says that atoms are made out of 3 different subatomic particles (protons, neutrons and electrons). We will explore the nuclear model in more detail in the next section.
Development of the Plum Pudding Model
In 1804, John Dalton thought that atoms were tiny spheres that could not be divided. Each of the different elements were made out of different spheres.
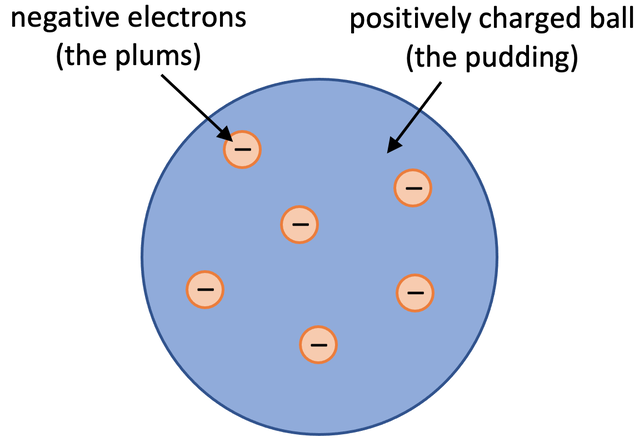
Then J Thomson undertook some experiments and discovered that atoms have negative charges (electrons). Thomson modified Dalton’s theory to say that atoms were a ball of positive charge with negative electrons embedded in the ball of positive charge. His theory was referred to as the plum pudding model. In the plum pudding model, the plums are the electrons and the pudding is the positively charged ball. A diagram of the plum pudding model is shown below and from the diagram, you can see that the atom is a positive ball with negative electrons inside.
In 1804, John Dalton thought that atoms were tiny spheres that could not be divided. Each of the different elements were made out of different spheres.
Then J Thomson undertook some experiments and discovered that atoms have negative charges (electrons). Thomson modified Dalton’s theory to say that atoms were a ball of positive charge with negative electrons embedded in the ball of positive charge. His theory was referred to as the plum pudding model. In the plum pudding model, the plums are the electrons and the pudding is the positively charged ball. A diagram of the plum pudding model is shown below and from the diagram, you can see that the atom is a positive ball with negative electrons inside.
Rejection of the Plum Pudding Model
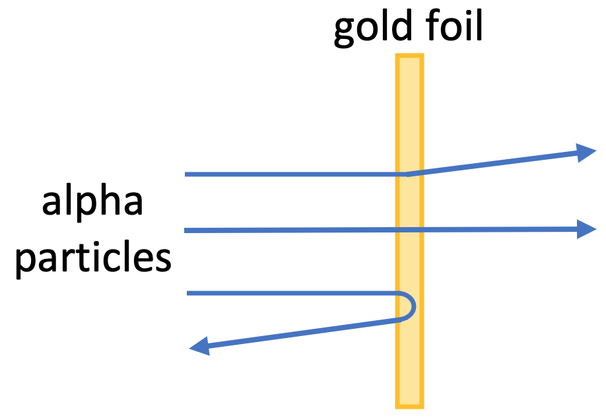
In the early 20th centaury, Rutherford and Marsden undertook an experiment that proved that the plum pudding theory was incorrect. The experiment involved firing alpha particles at a very thin sheet of gold foil. An alpha particle is a helium nuclei (it has 2 protons, 2 neutrons and 0 electrons). Alpha particles are positively charged because they have 2 protons and 0 electrons.
During the experiment, the scientists monitored the passage of the alpha particles through the gold foil. The plum pudding model predicted that the majority of the alpha particles would go straight through the sheet of gold foil or only be slightly deflected.
However, Rutherford and Marsden observed that:
These observations contradicted the predictions from the plum pudding model.
In the early 20th centaury, Rutherford and Marsden undertook an experiment that proved that the plum pudding theory was incorrect. The experiment involved firing alpha particles at a very thin sheet of gold foil. An alpha particle is a helium nuclei (it has 2 protons, 2 neutrons and 0 electrons). Alpha particles are positively charged because they have 2 protons and 0 electrons.
During the experiment, the scientists monitored the passage of the alpha particles through the gold foil. The plum pudding model predicted that the majority of the alpha particles would go straight through the sheet of gold foil or only be slightly deflected.
However, Rutherford and Marsden observed that:
- most of the alpha particles passed straight through as expected
- some of the alpha particles passed through the foil and were deflected more than expected
- a small number of the alpha particles were deflected back from where they came from, which was not expected
These observations contradicted the predictions from the plum pudding model.
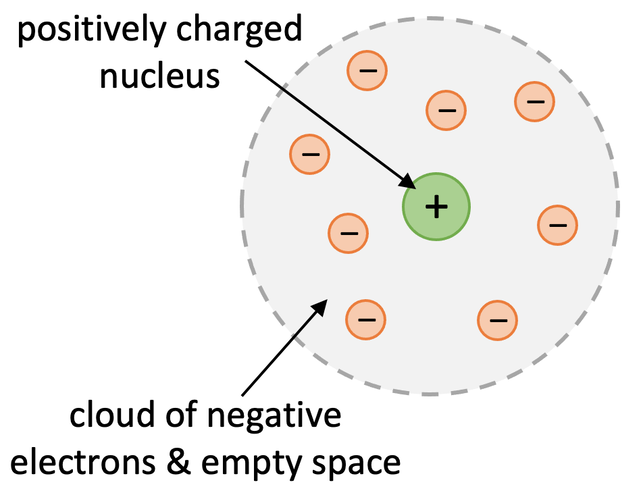
Rutherford and Marden tried to work out what was happening and concluded that most of the mass of the atom was concentrated in the centre of the atom in a tiny nucleus which was positively charged. They also concluded that the positively charged nucleus was surrounded by a cloud of electrons and a lot of empty space.
Most of the alpha particles passed through the empty space in the gold atoms in the gold foil and therefore continued to travel straight. The positively charged nucleus of the gold atoms explains why some of the alpha particles were deflected more than expected. This is because the alpha particles are positive and so is the small nuclei of the gold atoms. Whenever an alpha particle passed near to the nucleus of a gold atom, it would be deflected because the same charges repel one another; the positively charged gold nuclei and positively charged alpha particle would repel each other, which will result in the alpha particle having its pathway changed. Whenever the alpha particle was fired directly at the nuclei of a gold atom, the alpha particle would be deflected backwards towards the source that was emitting the alpha particles for the same reason; the positively charged gold nuclei would repel the positively charged alpha particle.
Bohr’s Nuclear Model
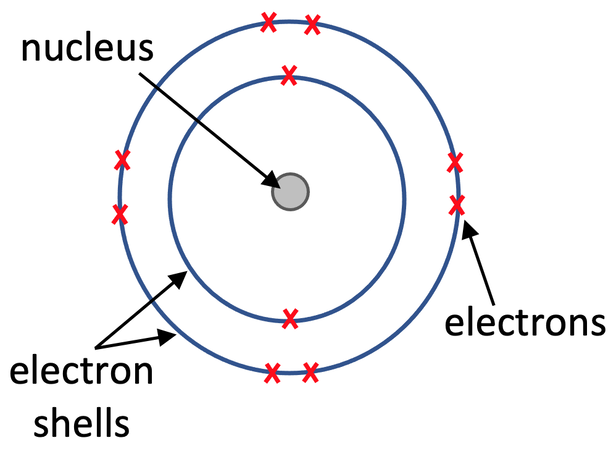
Scientists thought that the electrons that are in the cloud around the nucleus would be attracted to the positively charged nucleus because opposite charges attract. This would cause the atom to collapse. Bohr’s nuclear model suggested that the electrons orbit the nucleus in shells. Each of the shells were at fixed distances from the positively charged nucleus.
Scientists thought that the electrons that are in the cloud around the nucleus would be attracted to the positively charged nucleus because opposite charges attract. This would cause the atom to collapse. Bohr’s nuclear model suggested that the electrons orbit the nucleus in shells. Each of the shells were at fixed distances from the positively charged nucleus.
Bohr’s theoretical model agreed with experimental observations.
Further Experiments
Later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles. Each of these smaller particles have the same positive charge and they are known as protons.
The experimental work of James Chadwick provided the evidence to show that the nucleus of an atom contains neutral particles that are now known as neutrons. Chadwick discovered this around 20 years after it was widely accepted that atoms contain a nucleus. This model is extremely close to the nuclear model that we accept today. We will be looking at the nuclear model in more detail in the next section.
Later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles. Each of these smaller particles have the same positive charge and they are known as protons.
The experimental work of James Chadwick provided the evidence to show that the nucleus of an atom contains neutral particles that are now known as neutrons. Chadwick discovered this around 20 years after it was widely accepted that atoms contain a nucleus. This model is extremely close to the nuclear model that we accept today. We will be looking at the nuclear model in more detail in the next section.