Back to C1 Home
C1 L) Metals & Non-Metals
C1 L) Metals & Non-Metals
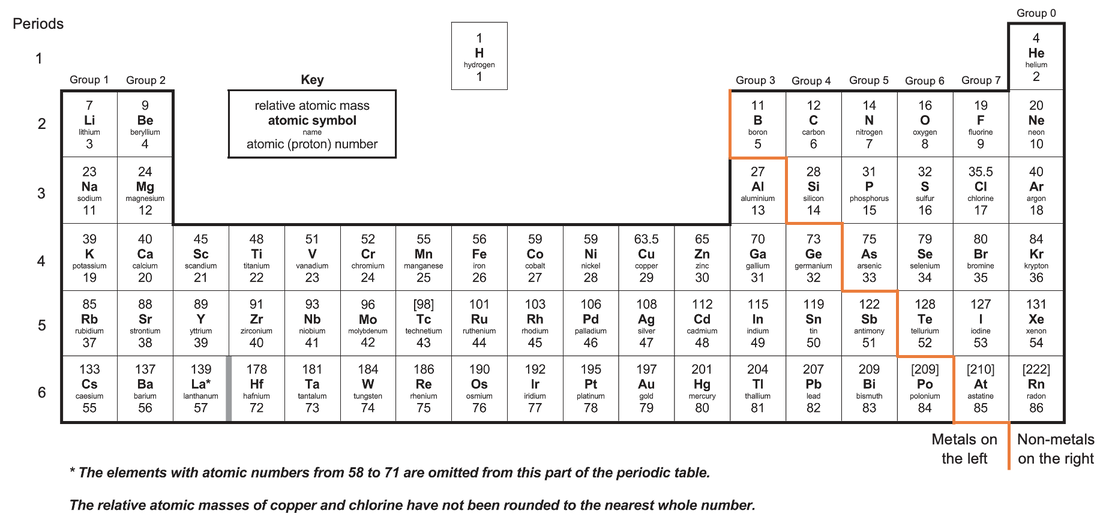
Elements that react to form positive ions are metals, and elements that do not react to form positive ions are non-metals. Metals are towards the left and bottom of the periodic table. Non-metals are towards the right and top of the periodic table. The periodic table with the metal and non-metal split is shown below.
From looking at the periodic table, you can see that most of the elements are metals.
Reactions of Metals & Non-Metals
Atoms want to react so that their outer shells are full. Atoms can fill their outer shells by losing, gaining or sharing electrons.
Metals are towards the left and bottom of the periodic table. Metals towards the left of the periodic table have few electrons in their outermost shell; the metals in group 1 only have 1 electron in their outermost shell, and the metals in group 2 only have 2 electrons in their outermost shell. Also, metals towards the bottom of the periodic table have electrons in an outer shell that is quite far away from the positively charged nucleus (the nucleus of atoms are positively charged because protons are found in the nucleus). For both the metals on the left of the periodic table (group 1 and group 2) and the metals at the bottom of the periodic table, not much energy is needed to remove these electrons in their outermost shells. This means that it is quite easy for metals to lose their electrons and react to form positive ions (electrons are negatively charged, so when they are removed/ lost, the metal will have more protons than electrons, resulting in the metal becoming positively charged – a positive ion).
Non-metals are towards the right and top of the periodic table. For the non-metals on the right of the periodic table, there are a lot of electrons in their outermost shell. In order for these non-metals to become positive ions, they would need to lose many electrons, which is unlikely. Also, for the non-metals towards the top of the periodic table, the electrons on the outermost shell are on a closer shell to the positively charged nucleus. This means that there is a strong force of attraction between the electrons on the outermost shell and the positively charged nucleus, which makes it hard for these non-metals to lose the electrons in their outermost shell and become positive ions. For all non-metals, it is easier for them to either gain electrons or share electrons to obtain full outer shells.
Atoms want to react so that their outer shells are full. Atoms can fill their outer shells by losing, gaining or sharing electrons.
Metals are towards the left and bottom of the periodic table. Metals towards the left of the periodic table have few electrons in their outermost shell; the metals in group 1 only have 1 electron in their outermost shell, and the metals in group 2 only have 2 electrons in their outermost shell. Also, metals towards the bottom of the periodic table have electrons in an outer shell that is quite far away from the positively charged nucleus (the nucleus of atoms are positively charged because protons are found in the nucleus). For both the metals on the left of the periodic table (group 1 and group 2) and the metals at the bottom of the periodic table, not much energy is needed to remove these electrons in their outermost shells. This means that it is quite easy for metals to lose their electrons and react to form positive ions (electrons are negatively charged, so when they are removed/ lost, the metal will have more protons than electrons, resulting in the metal becoming positively charged – a positive ion).
Non-metals are towards the right and top of the periodic table. For the non-metals on the right of the periodic table, there are a lot of electrons in their outermost shell. In order for these non-metals to become positive ions, they would need to lose many electrons, which is unlikely. Also, for the non-metals towards the top of the periodic table, the electrons on the outermost shell are on a closer shell to the positively charged nucleus. This means that there is a strong force of attraction between the electrons on the outermost shell and the positively charged nucleus, which makes it hard for these non-metals to lose the electrons in their outermost shell and become positive ions. For all non-metals, it is easier for them to either gain electrons or share electrons to obtain full outer shells.
The Characteristics
Metals will form metallic bonds (click here for more information on metallic bonding). This results on metals tending to have the following properties:
Non-metals tend to have the following properties:
Metals will form metallic bonds (click here for more information on metallic bonding). This results on metals tending to have the following properties:
- Strong – hard to break
- Malleable – can be bent into different shapes
- Good conductors of heat and electricity
- High melting and boiling points
- High densities
Non-metals tend to have the following properties:
- Brittle/ not strong – easy to break
- Dull looking
- Very poor conductors of heat and they generally don’t conduct electricity
- Lower melting and boiling points – they are usually gases at room temperature
- Low densities