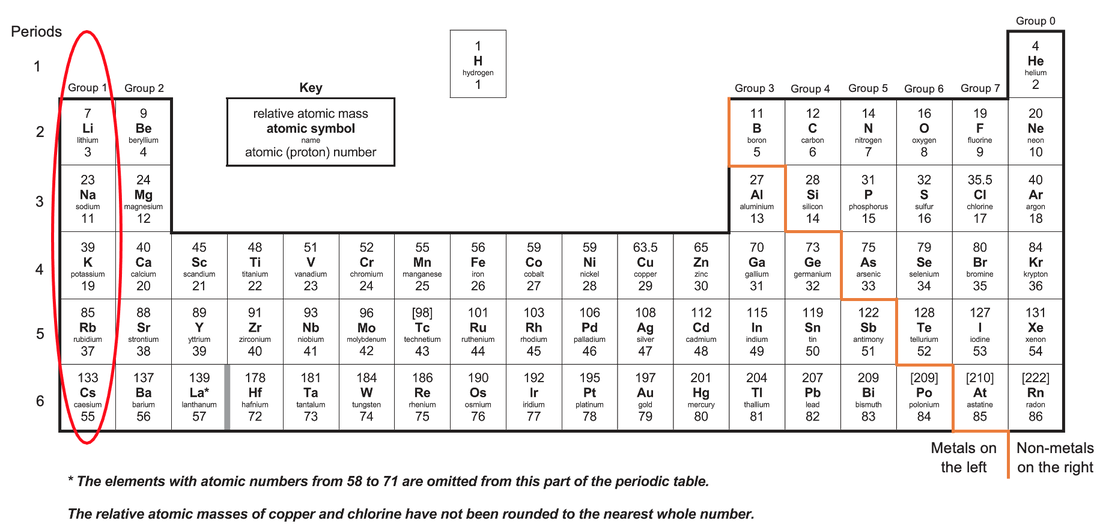
C1 M) Group 1 Elements
The elements in group 1 all have similar properties. They have low melting and boiling points for metals, a low density and are very soft (they can be cut with a knife).
Group 1 elements have one electron in their outermost electron shell, which they will want to lose so that they have a full outer shell of electrons. They lose this single electron by reacting with a non-metal to form ionic compounds. The ionic compounds that are formed are usually white and they dissolve in water to form a colourless solution. The reaction between a group 1 metal and a non-metal works by the group 1 metal giving one electron to the non-metal. This results in the group 1 metal becoming positively charged as it has given/ lost an electron, and the non-metal becoming negatively charged as it has gained an electron. The group 1 elements will become 1+ ions (Li+, Na+, K+ etc).
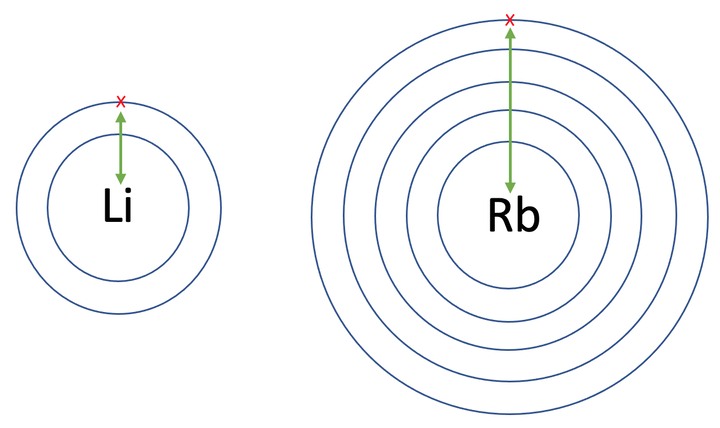
Group 1 elements are very reactive because they only have to lose one electron to obtain a full outer shell of electrons and become stable. The reactivity of the group 1 elements increases as we go down the group 1 column. For example, lithium is the least reactive, then sodium, potassium, rubidium, caesium and francium is the most reactive. The reactivity increases as we go down the group 1 column because the single electron that a lower down group 1 element wants to lose is on an electron shell that is further away from the positively charged nucleus. This means that there is a weaker force of attraction between the positively charged nucleus and the electron that further down group 1 elements are trying to lose. This weaker force of attraction means that less energy is required to remove the electron on the outmost shell of a further down group 1 element, which results in the reactivities increasing as we go down the group 1 column. To show that this is the case, I have drawn the shells for lithium and rubidium.
Therefore, as we go down the group 1 column, the electron that the group 1 element is trying to lose is on a shell further away from the positively charged nucleus, which means that there is a weaker force of attraction between the nucleus and the electron. This means that less energy is required to take this electron away/ for it to react, thus resulting in the reactivities of group 1 elements increasing as we go down the group 1 column.
Here is a summary of the property trends of the group 1 elements:
- The melting and boiling points decrease as we go down the group 1 elements; lithium has the highest melting and boiling point, and francium has the lowest melting and boiling point.
- The densities increase as we go down the group 1 elements; lithium has the lowest density, and francium has the highest density.
- The reactivities increase as we go down the group 1 elements; lithium is the least reactive, and francium is the most reactive.
Group 1 metals are so reactive that they are stored in oil (if they weren’t stored in oil, they would react with the oxygen in air). If we cut a group 1 metal, the part of the group 1 metal that is on show/ exposed will start to react with oxygen to form a dull metal oxide; the metal will go from shinny after it has been cut to a dull colour (which is the metal oxide forming). This process will be extremely quick, and you would be able to see the metal change from shinny to dull in a matter of seconds/ minutes.
All of the group 1 metals react with oxygen to form metal oxides.
Lithium reacts with oxygen to form lithium oxide. The equation for this is shown below:
Sodium will react with oxygen to form a mixture of sodium oxide (Na2O) and sodium peroxide (Na2O2).
Potassium will react with oxygen to form a mixture of potassium peroxide (K2O2) and potassium superoxide (KO2).
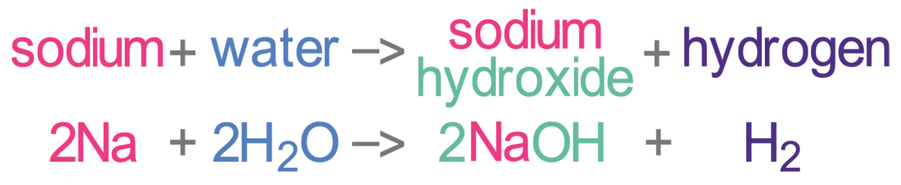
Group 1 metals react vigorously with water to produce a metal hydroxide and hydrogen gas. The hydroxides that they produce dissolve into the water resulting in the water becoming alkaline (hence why they are called the alkali metals). The hydroxide that is produced is dependent on the group 1 metal that is used in the reaction. For example, sodium reacts with water to produce sodium hydroxide and hydrogen.
The same reactions will take place for all of the other group 1 elements. Lithium reacts with water to produce lithium hydroxide and hydrogen.
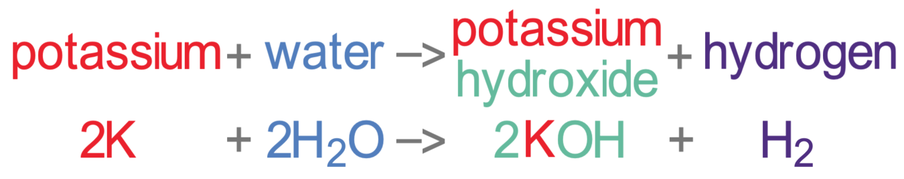
Potassium reacts with water to produce potassium hydroxide and hydrogen.
The reactions of the group 1 elements become more aggressive as we move down the group 1 column:
- When lithium (the least reactive group 1 metal) reacts with water, it moves around the surface of the water and fizzes. The fizzing is the hydrogen gas being produced.
- When sodium reacts with water, it moves around the surface of water and produces a significant amount of heat, which results in the sodium melting.
- When potassium reacts with water, it moves around the surface of water and fizzes. The reaction will also produce vast quantities of heat, which causes the hydrogen gas that is produced by the reaction to ignite (we will observe a flame). The heat also causes the potassium to melt.
- Group 1 metals further down (like rubidium and caesium) react so violently with water that they explode when they come into contact with water (click here to see a YouTube video of some of the further down group 1 elements reacting with water).
When the group 1 elements react with water, they produce hydrogen. We can test for the presence of hydrogen by catching some of the gas that is produced in a test tube. We then place a lit splint into the test tube. If hydrogen is present, we will hear a squeaky pop.
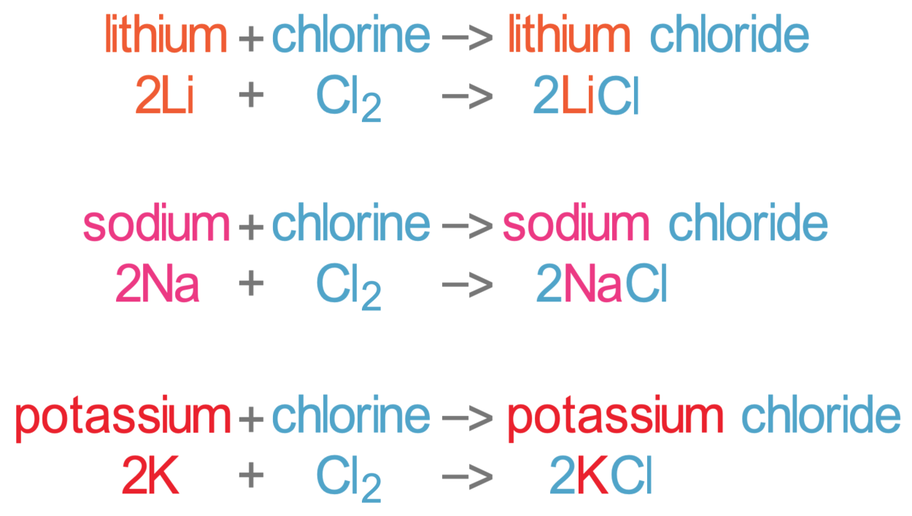
Group 1 metals react vigorously when heated with chlorine gas. The reactions become more violent if the group 1 metal involved is further down the group 1 column. The reaction produces a metal chloride, which are white salts.
The reactions for the first 3 group 1 metals (lithium, sodium and potassium) are shown below:
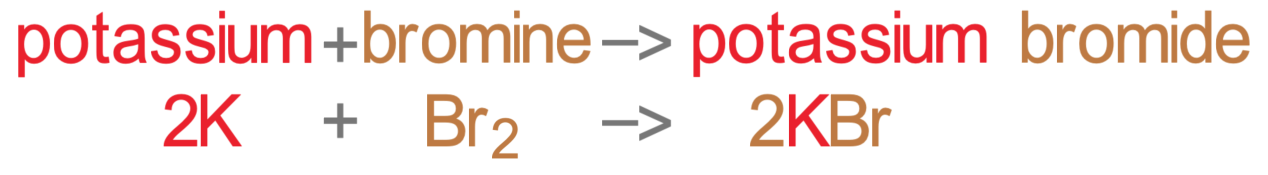
The reactions above will essentially be the same with any other group 7 element. For example, if we reacted potassium with bromine, we would obtain potassium bromide (rather than potassium chloride). Here is the equation for the reaction of potassium and bromine.