Back to C1 Home
C1: Quiz 1
C1: Quiz 1
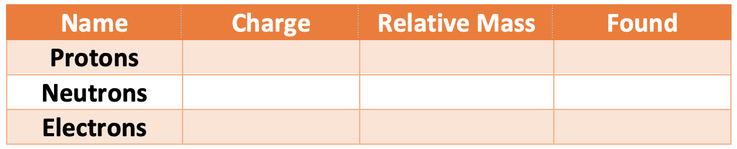
1) The three different subatomic particles are shown in the table below.
Fill in the missing values in the table.
2) What is the radius of an atom in nanometres?
3) How many times smaller is the radius of the nucleus of an atom compared to the radius of the whole atom?
4) What is the radius of the nucleus of an atom in metres?
5) How many electrons can the…
a) first shell hold?
b) second shell hold?
c) third shell hold?
6) Which electron shell will the electrons fill first; the closest shell to the nucleus or the furthest shell away from the nucleus?
7) The equations/ inequalities below show the number of protons and electrons in some atoms. Say whether the atoms are neutrally charged, positively charged or negatively charged.
a) protons < electrons
b) electrons < protons
a) protons = electrons
2) What is the radius of an atom in nanometres?
3) How many times smaller is the radius of the nucleus of an atom compared to the radius of the whole atom?
4) What is the radius of the nucleus of an atom in metres?
5) How many electrons can the…
a) first shell hold?
b) second shell hold?
c) third shell hold?
6) Which electron shell will the electrons fill first; the closest shell to the nucleus or the furthest shell away from the nucleus?
7) The equations/ inequalities below show the number of protons and electrons in some atoms. Say whether the atoms are neutrally charged, positively charged or negatively charged.
a) protons < electrons
b) electrons < protons
a) protons = electrons