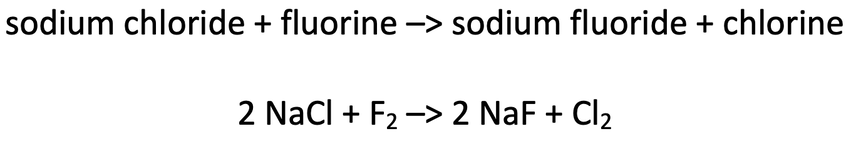C1: Quiz 14 – Answers
1)
a) The most reactive halogen is fluorine
b) The reactivity of halogens decreases as we go down the group 7 elements
2)
a) sodium bromide + chlorine –> sodium chloride + bromine
b) 2 NaBr + Cl2 –> 2 NaCl + Br2
c) Displacement reaction
3)
a) sodium iodide + fluorine –> sodium fluoride + iodine
b) 2 NaI + F2 –> 2 NaF + I2
4)
a) Yes. Chlorine is more reactive than bromine as it is higher up in the group 7 column in the periodic table. This means that chlorine will displace bromine and a reaction will take place
b) No. Iodine is less reactive than bromine as it is further down in the group 7 column in the periodic table. This means that iodine will be unable to displace bromine, thus meaning that a reaction will not take place
5)
a) Oxidation is the loss of electrons
b) Reduction is the gain of electrons
OIL RIG stands for; Oxidation Is Loss, Reduction Is Gain
6)
a)
i) Cl-, -1 or 1-
ii) 0, Cl or no charge
iii) lost 1 electron
iv) chlorine has been oxidised
b)
i) 0, F or no charge
ii) F-, -1 or 1-
iii) gained 1 electron
iv) fluorine has been reduced
c) 2 Cl- + F2 –> Cl2 + 2 F-
7)
a) reduced
b) oxidised
8)
Word: potassium iodide + bromine –> potassium bromide + iodine
Balanced chemical equation: 2 KI + Br2 –> 2 KBr + I2
b) Iodine is oxidised as it lost an electron
c) Bromine is reduced as it gained an electron
d) 2 I- + Br2 –> I2 + 2 Br-
9)
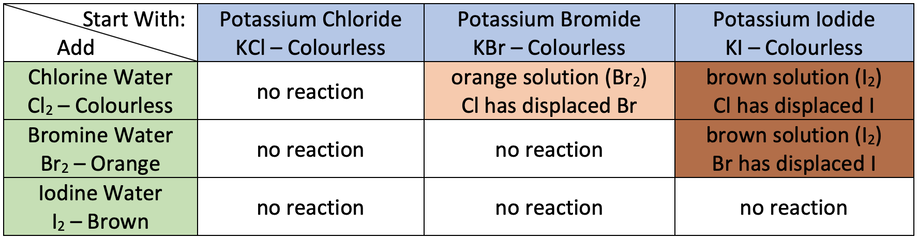
a) The experiment starts by placing a small amount of a halide salt solution in a test tube. We then add a few drops of a halogen solution to the test tube and shake gently. There are two different outcomes that we can observe:
- If we observe a colour change, a reaction has taken place; the halogen in the halogen solution has displaced the halogen in the halide salt solution. This tells us that the halogen in the halogen solution is a more reactive halogen than the halogen in the halide salt solution.
- If we do not observe a colour change, no reaction has taken place. This tells us that the halogen in the halide salt solution is more reactive than the halogen in the halogen solution.
b)
1)
a) Which is the most reactive halogen?
b) Describe the trend in the reactivities of the halogens. You do not need to explain why.
2) Sodium bromide reacts with chlorine.
a) Write the word equation for this reaction.
b) Write the balanced chemical equation for this reaction.
c) Give the name for this type of reaction.
3) Sodium iodide reacts with fluorine.
a) Write the word equation for this reaction.
b) Write the balanced chemical equation for this reaction.
4)
a) Will potassium bromide react with chlorine? Explain your answer.
b) Will potassium bromide react with iodine? Explain your answer
5) Use OIL RIG to help you answer this question.
a) In terms of electrons, is oxidation the loss or gain of electrons?
a) In terms of electrons, is reduction the loss or gain of electrons?
6) Fluorine will react with sodium chloride to produce sodium fluoride and chlorine. The word equation and balanced symbol equation for this reaction is shown below.
i) What is the charge on the chloride ion on the left side of the reaction?
ii) What is the charge on the chlorine on the right side of the reaction?
iii) Has chlorine gained or lost an electron?
iv) Has chorine been oxidised or reduced?
b)
i) What is the charge on the fluorine on the left side of the reaction?
ii) What is the charge on the fluoride ion on the right side of the reaction?
iii) Has fluorine gained or lost electrons?
iv) Has fluorine been oxidised or reduced?
c) Write the ionic equation for this reaction.
7) Answer the following questions with oxidised or reduced.
a) In a halogen displacement reaction, the displacing halogen is _______.
b) In a halogen displacement reaction, the displaced halogen is _______.
8) Potassium iodide reacts with bromine water.
a) Write the word equation and the balanced symbol equation for this reaction.
b) Which substance is oxidised in this reaction? Explain your answer.
c) Which substance is reduced in this reaction? Explain your answer.
d) Write the ionic equation for this reaction.
9)
a) Describe how we can carry out an experiment to investigate the reactivities of different halogens.
b) Fill in the table below for what we will observe.