Back to C1 Home
C1: Quiz 8
C1: Quiz 8
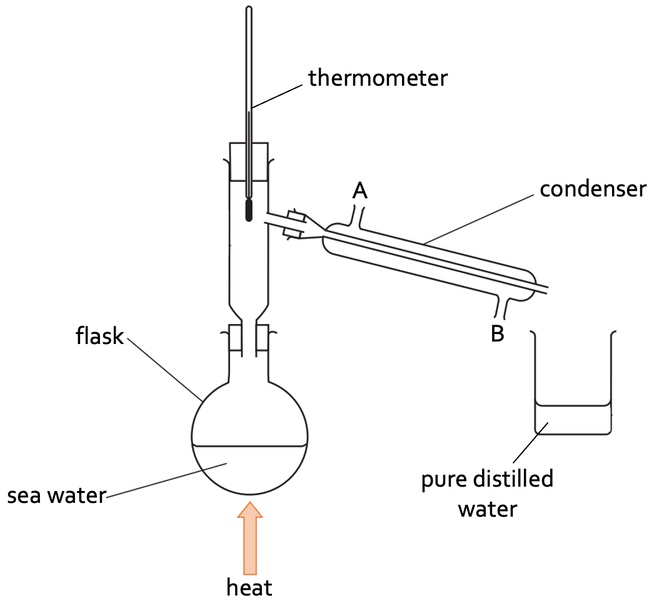
1) The diagram below shows how we can separate sea water to obtain pure distilled water.
a) Describe how we can use the apparatus above to obtain pure distilled water.
b) The condenser has two holes in it where cold water from a tap enters and leaves the condenser; these holes are labelled as A and B on the above diagram.
i) Through which hole does cold water enter the condenser?
ii) Through which hole does cold water leave the condenser?
2) When would we use fractional distillation rather than simple distillation?
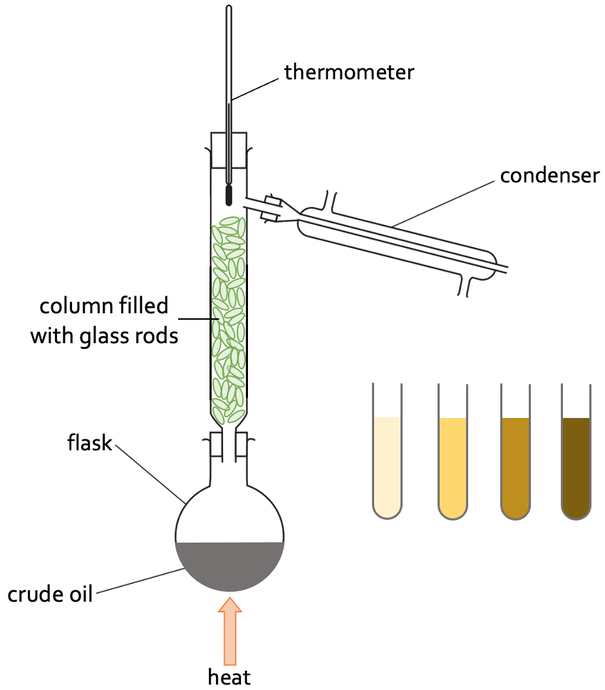
3) The set up for fractional distillation of crude oil is shown below.
b) The condenser has two holes in it where cold water from a tap enters and leaves the condenser; these holes are labelled as A and B on the above diagram.
i) Through which hole does cold water enter the condenser?
ii) Through which hole does cold water leave the condenser?
2) When would we use fractional distillation rather than simple distillation?
3) The set up for fractional distillation of crude oil is shown below.
a) When completing fractional distillation, do we go from a low temperature to a high temperature, or from a high temperature to a low temperature? Explain your answer.
b)
i) Where is the column filled with glass rods the hottest?
ii) Where is the column filled with glass rods the coolest?
c) Describe the role that the long column filled with glass rods has on the process.
4) A student completes fractional distillation on a mixture of 3 liquids. The three liquids and their boiling points are:
a) When a student completes fractional distillation on the mixture containing these 3 liquids, which of the liquids will be:
i) the first fraction that she obtains? Explain your answer.
ii) the second fraction that she obtains? Explain your answer.
iii) the third fraction that she obtains? Explain your answer.
b) The student uses the same apparatus that is shown in question 3. What piece of equipment will she use to ensure that just one of the fractions will evaporate?
b)
i) Where is the column filled with glass rods the hottest?
ii) Where is the column filled with glass rods the coolest?
c) Describe the role that the long column filled with glass rods has on the process.
4) A student completes fractional distillation on a mixture of 3 liquids. The three liquids and their boiling points are:
- Butanol: 118°C
- Propanol: 97°C
- Pentanol: 138°C
a) When a student completes fractional distillation on the mixture containing these 3 liquids, which of the liquids will be:
i) the first fraction that she obtains? Explain your answer.
ii) the second fraction that she obtains? Explain your answer.
iii) the third fraction that she obtains? Explain your answer.
b) The student uses the same apparatus that is shown in question 3. What piece of equipment will she use to ensure that just one of the fractions will evaporate?