C2 A) Ionic Bonds
Ionic bonding is where a metal and a non-metal react together, resulting in the metal giving electron(s) to the non-metal. The giving and receiving of electrons results in the metal and non-metal becoming charged; they become ions. The metal has given electrons, which means that it will become a positive ion because it now has more protons than electrons (protons are positive and electrons are negative). The non-metal has received electrons, which means that it will become a negative ion as it now has more electrons than protons. The oppositely charged metal (positive) and non-metal (negative) are now strongly attracted to each other by electrostatic forces of attraction, and this attraction is the ionic bonds.
We are now going to have a look at some dot and cross diagrams to show how ionic compounds are formed. One of the atoms involved in the bonding will have electrons that are dots, and the other atom will have electrons that are crosses. The benefits of a dot and cross diagram is that the dots and crosses clearly show us which atom the electrons come from.
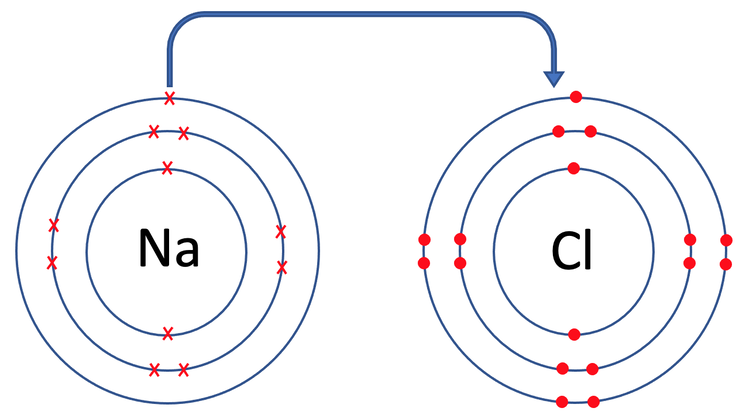
When we are drawing dot and cross diagrams for ionic bonds, it is best to draw the atoms prior to the bonding taking place, and then the atoms after the bonding has taken place. This is so that we can see how many electrons each of the atoms want to lose or gain.
Example 1 – Sodium Chloride
The first example that we are going to look at is sodium chloride, which is NaCl.
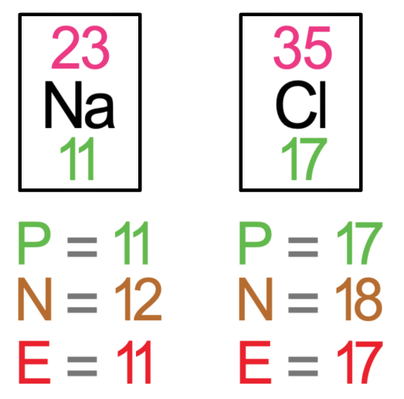
The first step is to draw the electronic shells for each of the atoms prior to the bonding taking place. The tiles in the periodic table for sodium and chlorine are shown below.
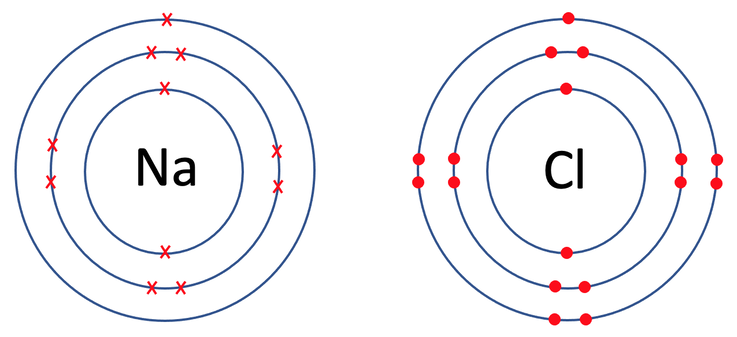
We now draw the electron shells for sodium and chloride before the ionic bonding has taken place. For the electron shells, the first shell takes 2 electrons, the second shell takes 8 electrons and the third shell also takes 8 electrons. Sodium has 11 electrons, which means that 2 will be in the first shell, 8 in the second shell and 1 in the third shell. Chlorine has 17 electrons, which means that 2 will be in the first shell, 8 in the second shell and 7 in the third shell. The electron shells for sodium and chlorine pre bonding are shown below.
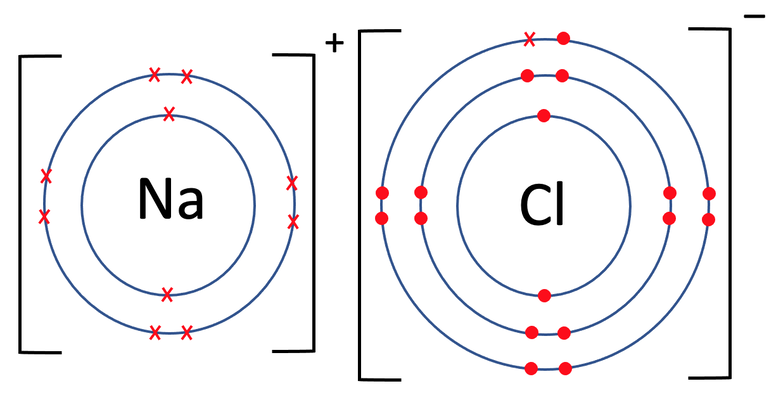
Both of the atoms involved are placed into brackets and we put the charge of each of the atoms on the top right outside the brackets. Sodium gave 1 electron, which results in it having a charge of 1+; we write it as Na+ (there is no need for the 1 in 1+, but you wouldn’t get marked down if you did include it). The chloride (chlorine after becoming an ion) has gained 1 electron, which results in it having a charge of 1-; we write it as Cl-. The charges along the top of the brackets should always add up to 0; here we have a positive (Na+) and a negative (Cl-), which adds to 0.
The oppositely charged positive sodium ion (Na+) and the negative chloride ion (Cl-) are now strongly attracted to each other by electrostatic forces of attraction, and this attraction is the ionic bond.
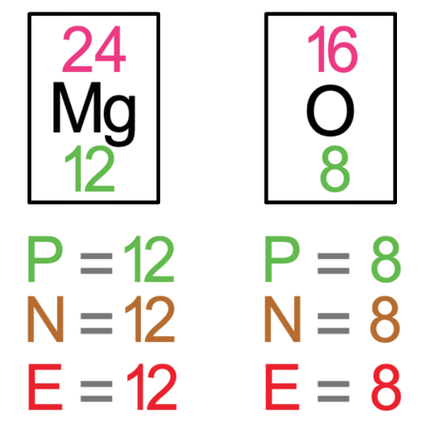
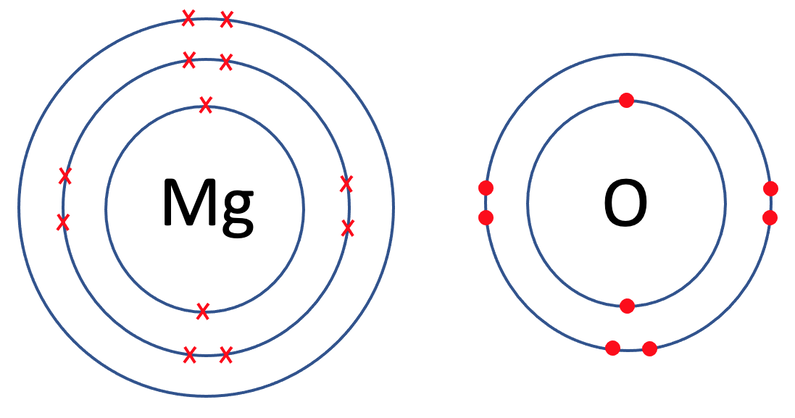
The next example that we are going to look at is magnesium oxide, which is MgO. Like the previous example, I am going to draw both of the atoms out prior to the bonding taking place. The tiles in the periodic table for magnesium and oxygen are shown below.
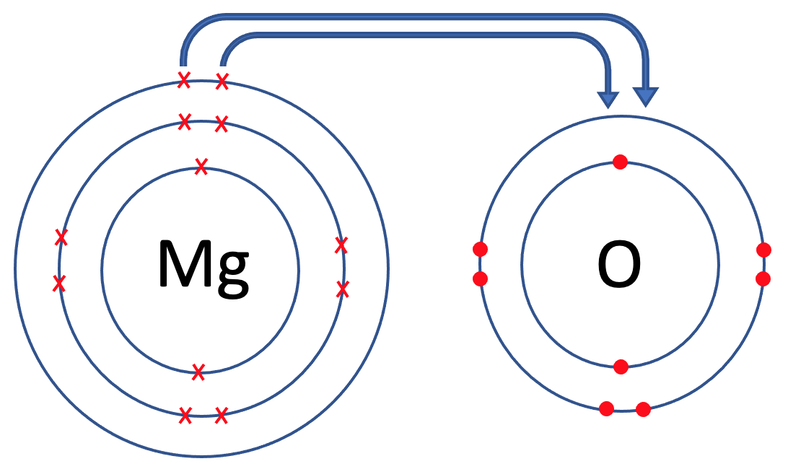
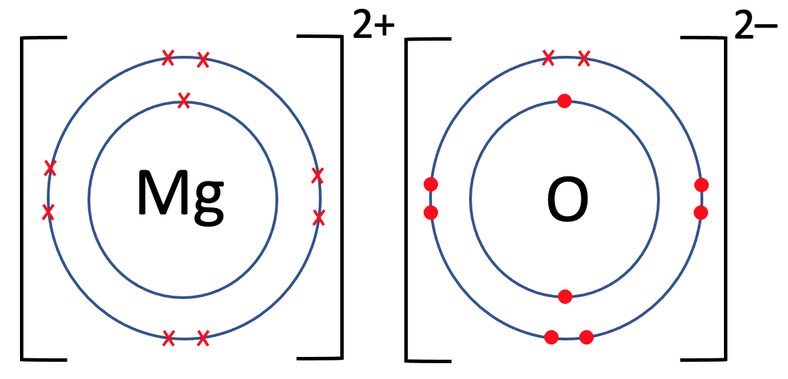
Both of the atoms involved are placed into brackets and we put the charge of each of the atoms on the top right outside the brackets. Magnesium gave 2 electrons, which results in it having a charge of 2+; we write it as Mg2+. The oxide (oxygen after becoming an ion) gained 2 electrons, which results in it having a charge of 2-; we write it as O2-. Like the example before, the charges along the top of the brackets will add up to 0. For magnesium oxide, we have 2+ and 2-, which does add up to 0.
The oppositely charged positive magnesium ion (Mg2+) and the negative oxide ion (O2-) are now strongly attracted to each other by electrostatic forces of attraction, and this attraction is the ionic bonds.
The previous 2 examples have only involved 2 atoms. We are now going to have an example where 3 atoms are involved.
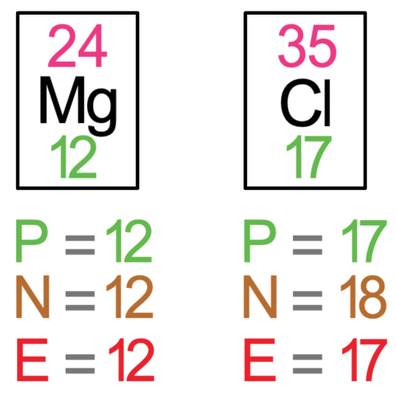
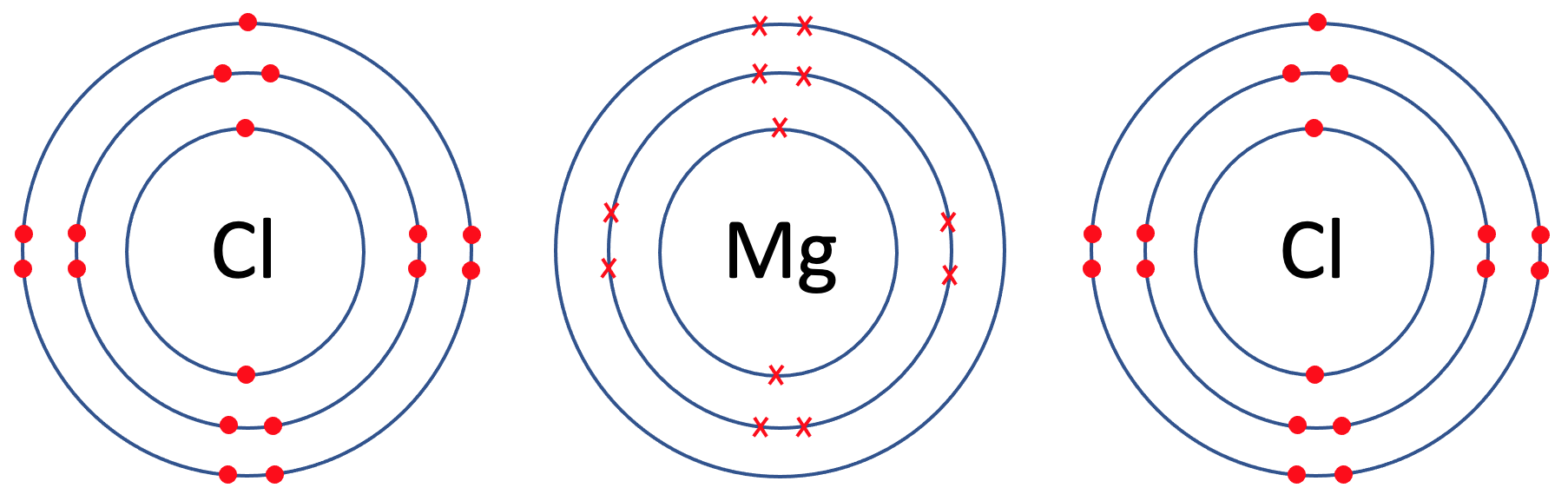
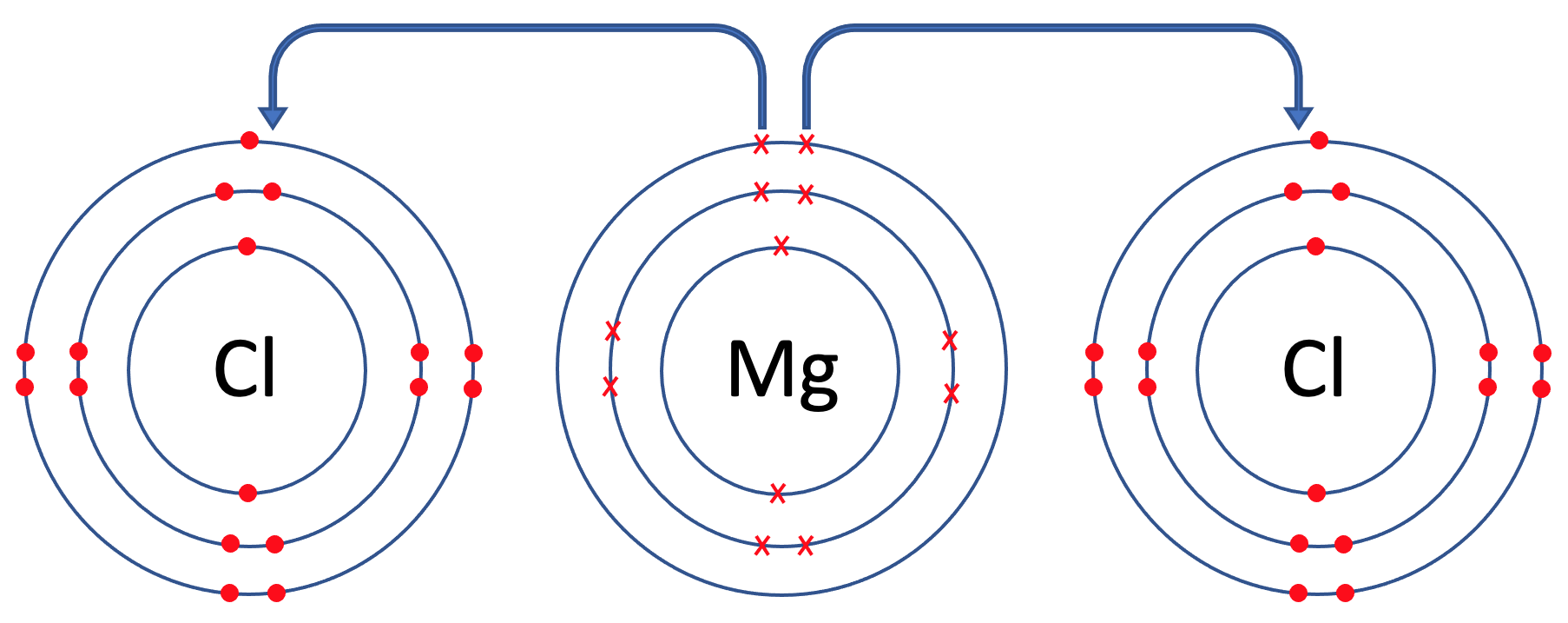
We are going to look at magnesium chloride, which has the formula MgCl2. In magnesium chloride, there is 1 magnesium atom and 2 chlorine atoms. The first step is to draw all of the atoms prior to the bonding taking place – we draw 1 magnesium atom and 2 chlorine atoms. The tiles for magnesium and chlorine are shown below.
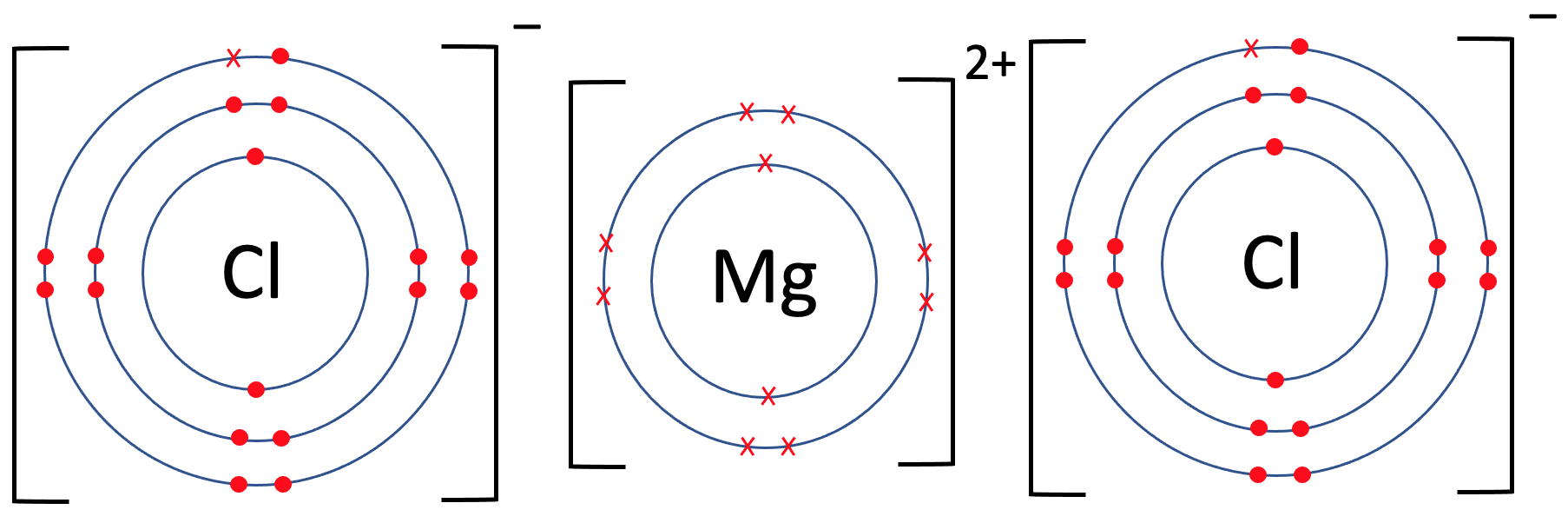
We now need to place the charges for all 3 of the atoms on the top right outside of the brackets. Magnesium gave 2 electrons, which means that it will have a charge of 2+; we write it as Mg2+. Both of the chlorides (chlorine after becoming an ion) gained 1 electron, which means that they will both have a charge of 1-; we write them as Cl-. Like the examples before, the charges along the top of the brackets will add up to 0; for these ionic bonds, the -, 2+ and -, does add up to 0.
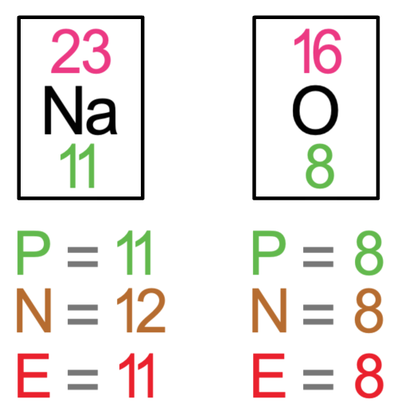
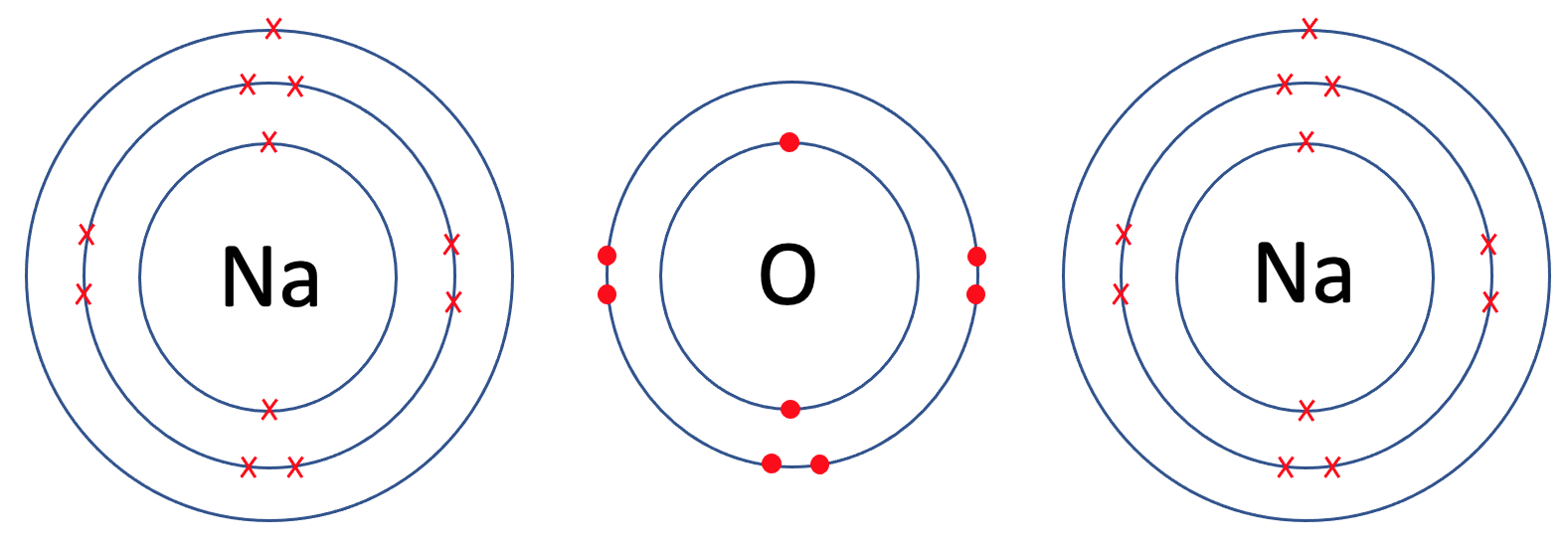
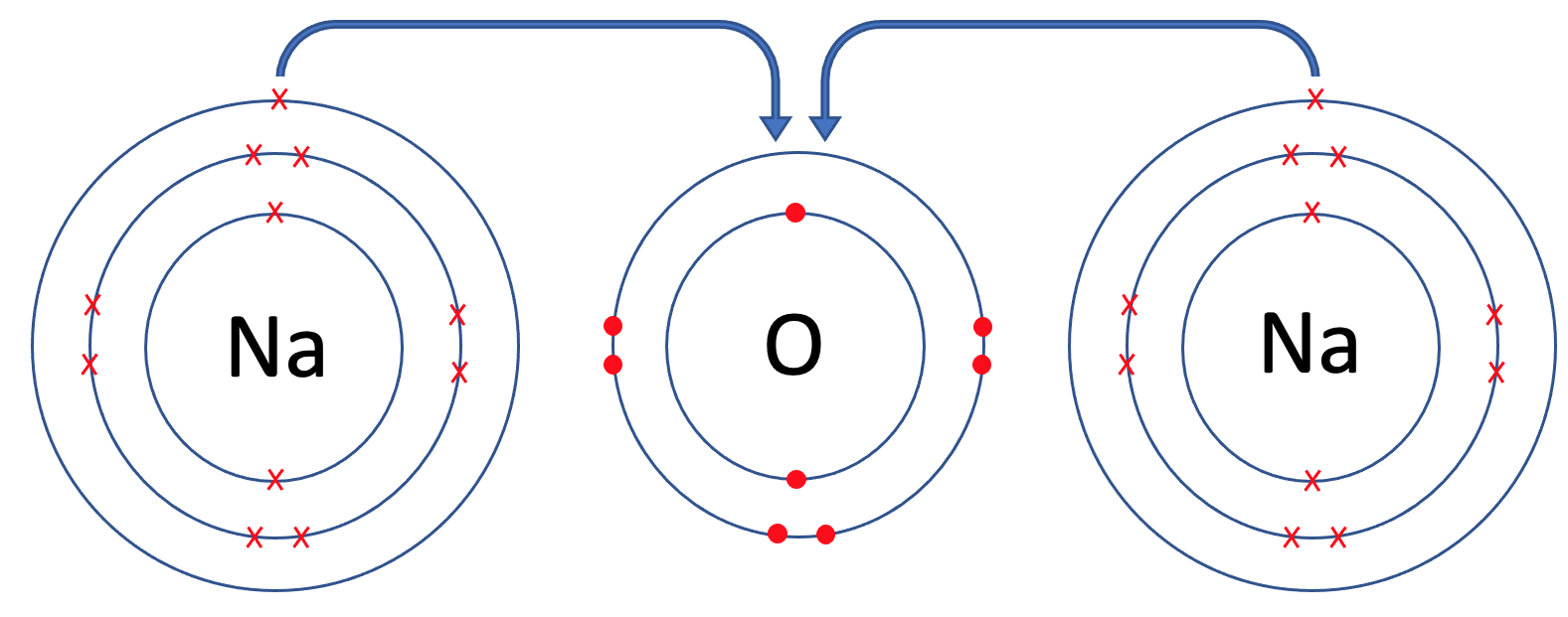
The final example that we are going to look at is sodium oxide, which is Na2O. In sodium oxide, there are 2 sodium atoms and one oxygen atom. Like all of the previous examples, the first step is to draw all of the atoms prior to the bonding taking place – we draw 2 sodium atoms and 1 oxygen atom. The tiles for sodium and oxygen are shown below.
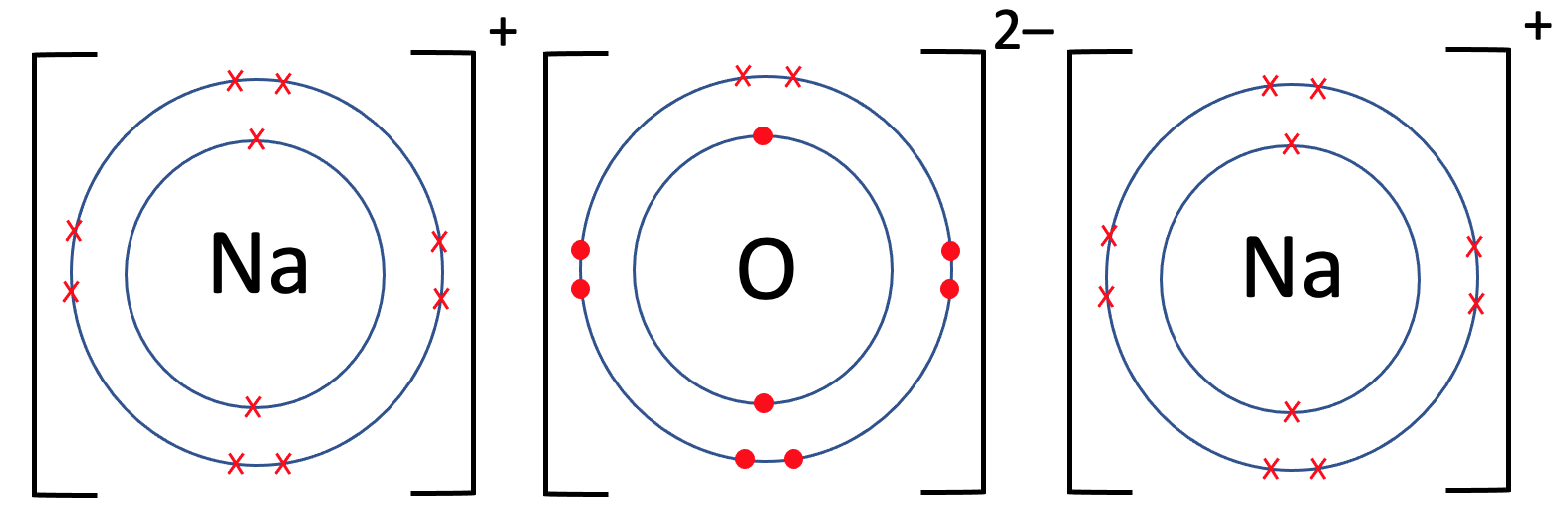
We now need to place the charges for all 3 of the atoms on the top right outside of the brackets. Each of the sodium atoms gave 1 electron, which means that they will have a charge of 1+; we write them as Na+. The oxide atom gained 2 electrons, which means that it will have a charge of 2-; we write it as O2-. All of the charges at the top of the brackets will add up to 0; for these ionic bonds, the +, 2– and +, does add up to 0.