C2 B) Ions from the Periodic Table
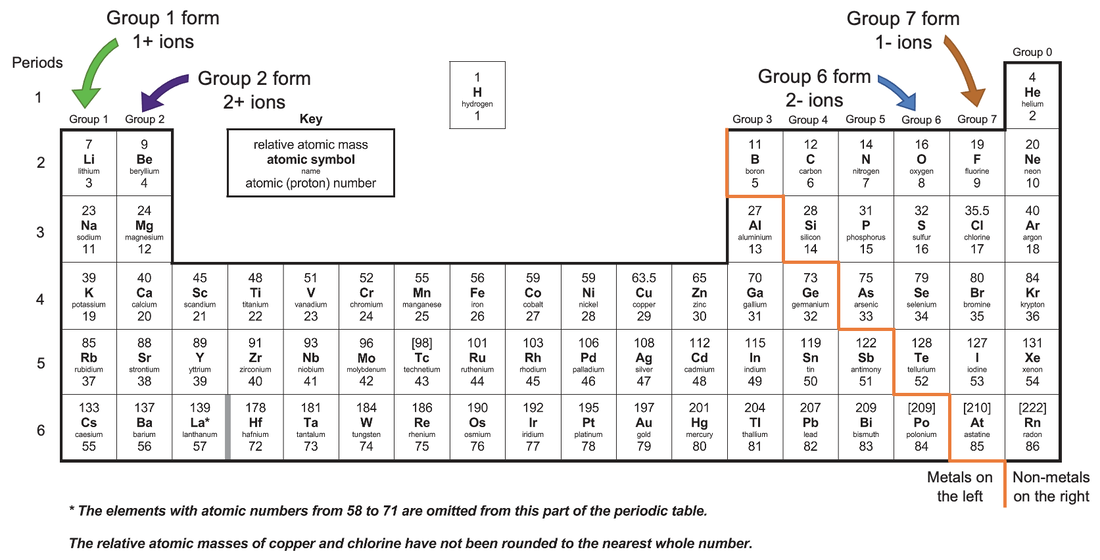
The groups in the periodic table tell us how many electrons the elements have in their outermost electron shell. Group 1, 2, 6 and 7 are the most likely elements to react and form ions. Group 1 and 2 elements are metals and they lose/ give electrons resulting in them becoming positive ions (they become cations). Group 6 and 7 elements are non-metals and they receive/ gain electrons resulting in them becoming negative ions (they become anions).
The different groups will lose or gain a different number of electrons.
Group 1 elements have 1 electron in their outermost shell. In order to have a full outer shell, they will want to lose this electron, which will result in them becoming positively charged by 1. For example, sodium will become Na+ after it has given 1 electron. The half equation for this is shown below.
Group 2 elements have 2 electrons in their outermost shell. In order to have a full outer shell, they will want to lose these 2 electrons, which will result in them becoming positively charged by 2. For example, magnesium will become Mg2+ after it has given 2 electrons. The half equation for this is shown below.
Group 7 elements have 7 electrons in their outermost shell. In order to have a full outer shell, they will want to gain 1 electron, which will result in them becoming negatively charged by 1. For example, fluorine will gain 1 electron to become a negative fluoride ion; it will be F-. The half equation for this is shown below.
Group 6 elements have 6 electrons in their outermost shell. In order to have a full outer shell, they will want to gain 2 electrons, which will result in them becoming negatively charged by 2. For example, oxygen will gain two electrons to become a negative oxide ion; it will be O2- after it has gained 2 electrons. The half equation for this is shown below.
I have added the ions that the different groups form to the periodic table below.
We are now going to have a look at working out the empirical formulas of ionic compounds by balancing out the charges. We balance out the charges by making sure that the ionic compound has no overall charge; the positive charge must be equal to/ cancelled out by the negative charge. The best way to understand how to find empirical formulas of ionic compounds is to go through a few examples.
Sodium Fluoride
What is the empirical formula of sodium fluoride?
Sodium fluoride is made out of sodium and fluorine atoms. We answer this question by finding the charges on both sodium and fluorine. Sodium is in group 1 in the periodic table, which means that it will lose 1 electron resulting in it having a 1 positive charge (Na+). Fluorine is in group 7 in the periodic table, which means that it will gain 1 electron resulting in it having a 1 negative charge (F-). The 1 positive charge from the sodium (Na+) and the 1 negative charge from fluorine (F-) balance each other out, thus meaning that we will have 1 sodium atom and 1 fluorine atom in sodium fluoride; the empirical formula for sodium fluoride is NaF.
What is the empirical formula of magnesium chloride?
Magnesium chloride is made out of magnesium and chlorine atoms. Magnesium is in group 2 in the periodic table, which means that it will lose 2 electrons resulting in it having a 2 positive charge (Mg2+). Chlorine is in group 7 in the periodic table, which means that it will gain 1 electron resulting in it having a 1 negative charge (Cl-). The overall charge for an ionic compound must be 0, which means that the charges from magnesium and chlorine must balance out. Therefore, as magnesium has a charge of 2+ (Mg2+), there must be 2 separate chlorines (Cl-) to give the ionic compound an overall charge of 0. This means that the empirical formula for magnesium chloride is MgCl2.
What is the empirical formula of potassium oxide?
Potassium oxide is made out of potassium and oxygen atoms. Potassium is in group 1 in the periodic table, which means that it will lose 1 electron resulting in it having a 1 positive charge (K+). Oxygen is in group 6 in the periodic table, which means that it will gain 2 electrons resulting in it having a 2 negative charge (O2-). The overall charge for an ionic compound must be 0, which means that the charges from potassium and oxygen must balance out. Therefore, as potassium has a charge of 1+ (K+), there must be 2 potassiums to balance out the 2- charge from the oxygen (O2-). This means that the empirical formula for potassium oxide is K2O.
What is the empirical formula of aluminium oxide?
Aluminium oxide is made out of aluminium and oxygen atoms. Aluminium is a metal and is in group 3 in the periodic table, which means that it will lose 3 electrons resulting in it having a 3 positive charge (Al3+). Oxygen is in group 6 in the periodic table, which means that it will gain 2 electrons resulting in it having a 2 negative charge (O2-). The overall charge for an ionic compound must be 0, which means that the charges from aluminium and oxygen must balance out. The balancing for aluminium oxide is slightly different because the positive and negative charges do not go into each other; the 3 positive charge from aluminium and the 2 negative charge from oxygen do not go into each other. The first number that 3 and 2 go into is 6 (the LCM is 6). This means that we can balance the charge of this ionic compound by having 6 positive charges and 6 negative charges. Each aluminium has a charge of 3+, which means that we can get 6 positive charges by having 2 aluminiums. Each oxide has a charge of 2-, which means that we can get 6 negative charges by having 3 oxides. Therefore, the empirical formula of aluminium oxide is Al2O3.