C2 C) Giant Ionic Compounds
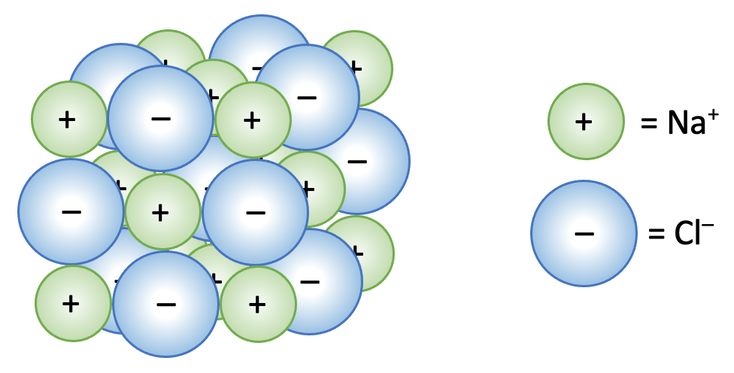
The positive sodium ions (Na+) and the negative chloride ions (Cl-) are alternating in all directions. The arrangement of the ions like this leads to extremely strong electrostatic forces of attraction between the oppositely charged ions. The strong electrostatic force of attraction between the ions takes a lot of energy to break/ be overcome, which is why ionic compounds have extremely high melting and boiling (we will look at this in more detail at the bottom of this section).
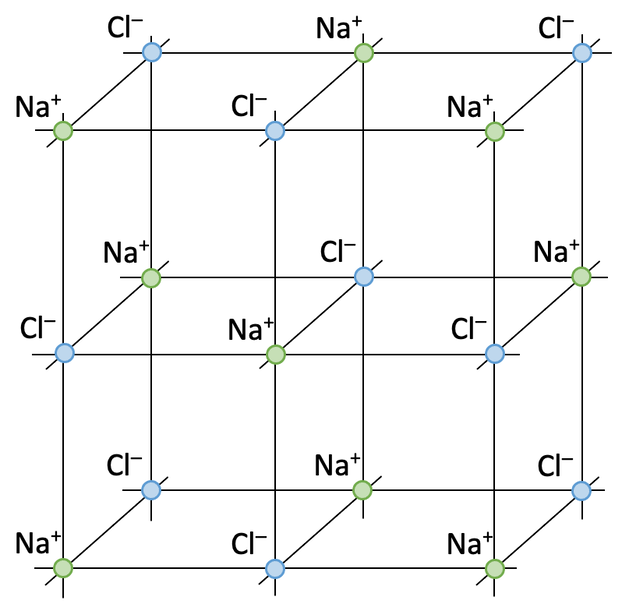
In the above diagram for sodium chloride, the relative sizes of the atoms are shown. The positive sodium ion is smaller than the negative chloride ion because sodium has 2 electron shells and chloride has 3 electron shells. Also, the diagram shows the 3-dimensional arrangement of all of the ions. A criticism of the above diagram is that it only shows the outside ions.
Ball & Stick Diagram
We could also show the arrangement of the ions by using a ball and stick diagram; this is shown below.
Ionic compounds have the following properties.
High Melting and Boiling Points
Ionic compounds have high melting and boiling points. This is because there are extremely strong electrostatic forces of attraction between all of the oppositely charged ions involved in the giant ionic compound. These strong forces of attraction take a lot of energy to be overcome/ broken, hence the high melting and boiling points.
The melting and boiling points of ionic compounds is influenced by the charges on the ions. In sodium chlorine, the sodium gives 1 electron to chlorine, which results in the sodium having a charge of 1+ and the chloride having a charge of 1-. Sodium chloride has a melting point of 801°C and a boiling point of 1,465°C.
In magnesium oxide, the magnesium gives 2 electrons to oxygen, which results in the magnesium having a charge of 2+ and the oxide having a charge of 2-. Magnesium oxide will have stronger electrostatic forces of attraction compared to sodium chloride; this is because the charges for magnesium oxide are 2+ and 2-, whereas the charges for sodium chloride are just 1+ and 1-. These stronger electrostatic forces of attraction for magnesium oxide require more energy to overcome/ break, thus meaning that magnesium oxide will have a higher melting and boiling point compared to sodium chloride. The melting point for magnesium oxide is 2,852°C and the boiling point is 3,600°C; both of these are higher than the respective melting and boiling points for sodium chloride.
Electricity
Ionic compounds are unable to conduct electricity when they are in solid form. This is because they do not have any delocalised electrons (electrons that can move) and the ions are unable to move as they are in their fixed positions in the solid.
Ionic compounds can conduct electricity when they are liquid because the ions are free to move in a liquid, which means that they will carry an electric current.
Some ionic compounds will dissolve in water (aqueous). When the ionic compounds dissolve in water, the ions separate and are free to move in the solution, thus meaning that they can carry an electric current.
So, ionic compounds will not conduct electricity in solid form, but they will conduct electricity if they are liquid or if they are dissolved in water.