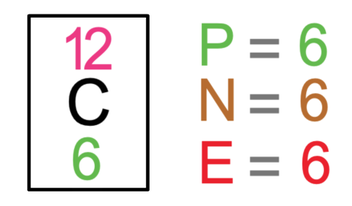Back to C2 Home
C2 F) Giant Covalent Structures
C2 F) Giant Covalent Structures
The atoms in giant covalent structures are held together by covalent bonds. Covalent bonds are when non-metals share pairs of electrons. Giant covalent structures contain lots of atoms and many covalent bonds. Giant covalent structures have the following properties:
Carbon is involved in many of the giant covalent substances that you need to know about for the exam. The tile for carbon from the periodic table is shown below.
- They have very high melting and boiling points. This is because there are many covalent bonds that need to be broken in order to change their state, which requires a large amount of energy, thus meaning that giant covalent structures have very high melting and boiling points.
- They are very strong because of the number of covalent bonds involved.
- Generally, they do not conduct electricity because there are no delocalised electrons or charged ions. There are a few exceptions to this rule, such as graphite and graphene that both conduct electricity.
Carbon is involved in many of the giant covalent substances that you need to know about for the exam. The tile for carbon from the periodic table is shown below.
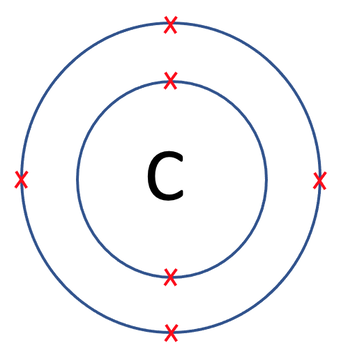
Carbon has 6 protons, 6 neutrons and 6 electrons. The electronic configuration for carbon will be 2 on the first shell and 4 on the second shell. Carbon and its electrons are drawn below.
Diamond
Diamond is made out of carbon, and each of the carbon atoms involved in diamond forms 4 single covalent bonds with 4 other carbon atoms. The carbons are joined in a very rigid structure and this makes diamond extremely hard. There are loads of covalent bonds in diamond, which take a lot of energy to break, thus meaning that diamond has a very high melting and boiling point. Diamond does not conduct electricity because there are no ions or any delocalised electrons (all of the 4 electrons in the outer shell for the carbon atoms are involved in covalent bonds meaning that there are no free/ delocalised electrons).
Diamond is made out of carbon, and each of the carbon atoms involved in diamond forms 4 single covalent bonds with 4 other carbon atoms. The carbons are joined in a very rigid structure and this makes diamond extremely hard. There are loads of covalent bonds in diamond, which take a lot of energy to break, thus meaning that diamond has a very high melting and boiling point. Diamond does not conduct electricity because there are no ions or any delocalised electrons (all of the 4 electrons in the outer shell for the carbon atoms are involved in covalent bonds meaning that there are no free/ delocalised electrons).
Diamond is used for cutting tools because it is extremely strong and has a very high melting point (a cutting tool needs a high melting point because the friction produced when the tool is cutting will result in the cutting tool becoming hot. If the tool had a low melting point, the tool would change shape, which would result in the cutting tool being less effective). If you are asked in the exam for a characteristic of diamond, do not say “shiny”.
Graphite & Graphene
Graphite is made out of many layers; a single layer is known as graphene. Graphite and graphene are made out of carbon atoms.
The carbon atoms in graphite form 3 single covalent bonds with 3 other carbon atoms, which creates sheets of carbon atoms that are arranged in hexagon. There are no covalent bonds between the layers of graphene in graphite. This results in there being a weak force of attraction between the layers, thus meaning that the layers can slide over one another. This is why graphite is sometimes referred to as soft and slippery. The sliding of the layers is why pencil makes a mark on a page. Graphite is also a good lubricating material (a lubricant is a substance that makes it easier for components to move past each other).
Graphite has a very high melting and boiling point because of the many covalent bonds that are involved in its structure. These bonds require a lot of energy to break, hence the very high melting and boiling point.
A carbon atom before it has bonded has 4 electrons in its outer shell. 3 of these electrons are used in the 3 single covalent bonds with other carbon atoms. This means that there is 1 electron that is delocalised (free to move), thus meaning that graphite conducts electricity. Graphite is also a good conductor of heat because of this delocalised electron.
Graphene is a single layer/ sheet of graphite. Graphene is extremely strong, light and only 1 atom thick (it is a 2D compound). Graphene can be added to composite materials to increase their strength without adding much mass (a composite material is a combination of two or more materials with different physical and chemical properties). Graphene is also able to conduct electricity because carbon only forms 3 single covalent bonds which means that 1 electron is delocalised. This is why graphene is used in electronics.
The carbon atoms in graphite form 3 single covalent bonds with 3 other carbon atoms, which creates sheets of carbon atoms that are arranged in hexagon. There are no covalent bonds between the layers of graphene in graphite. This results in there being a weak force of attraction between the layers, thus meaning that the layers can slide over one another. This is why graphite is sometimes referred to as soft and slippery. The sliding of the layers is why pencil makes a mark on a page. Graphite is also a good lubricating material (a lubricant is a substance that makes it easier for components to move past each other).
Graphite has a very high melting and boiling point because of the many covalent bonds that are involved in its structure. These bonds require a lot of energy to break, hence the very high melting and boiling point.
A carbon atom before it has bonded has 4 electrons in its outer shell. 3 of these electrons are used in the 3 single covalent bonds with other carbon atoms. This means that there is 1 electron that is delocalised (free to move), thus meaning that graphite conducts electricity. Graphite is also a good conductor of heat because of this delocalised electron.
Graphene is a single layer/ sheet of graphite. Graphene is extremely strong, light and only 1 atom thick (it is a 2D compound). Graphene can be added to composite materials to increase their strength without adding much mass (a composite material is a combination of two or more materials with different physical and chemical properties). Graphene is also able to conduct electricity because carbon only forms 3 single covalent bonds which means that 1 electron is delocalised. This is why graphene is used in electronics.
Fullerenes
Fullerenes are made out of carbon atoms. There are many different fullerenes that are made out of a different number of carbon atoms. Fullerenes can be cages, tubes or hollow balls.
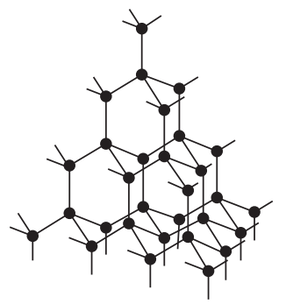
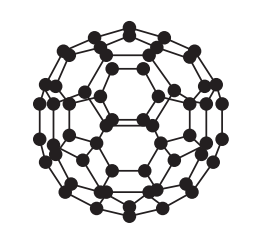
The first fullerene to be discovered was one called Buckminsterfullerene. It is a hollow sphere that is made out of 60 carbon atoms (C60). A diagram of Buckminsterfullerene is shown below.
The carbon atoms in fullerenes are joined together by covalent bonds. There are many covalent bonds involved in fullerenes, which means that fullerenes have high melting and boiling points. However, the melting and boiling points of fullerenes are lower than the melting and boiling points of graphite and diamond because there are fewer covalent bonds, thus meaning that less energy is required to break them, hence the lower melting and boiling points. Fullerenes are nanoparticles.
Fullerenes can be used to cage other molecules; the fullerenes go around the outside of another molecule. This is useful in medicine as we can trap a drug inside a fullerene and use the fullerene to deliver the drug to a specific part of the body. For example, nanoparticles have the potential to treat cancer by delivering chemotherapy drugs directly to the cancer cells. This will reduce the number of healthy body cells being destroyed by chemotherapy. Nanoparticles are currently being used in wound dressing. Manufacturers add silver nanoparticles to the fibres in the wound dressing and this gives the wound dressing antibacterial properties. The uses of fullerenes in medicine is currently being researched.
Fullerenes also have a very high surface area. We can attach a catalyst for a reaction to a fullerene, which makes the catalyst more effective (a catalyst is something that speed up the rate of a reaction without being used up in the reaction).
Fullerenes can be made into nanotubes that are very long in comparison with their diameter. The nanotubes have delocalised electrons, which means that they can conduct electricity and heat. They also have very high tensile strength, which means that they do not break when they are stretched. Nanotubes can be used in electronics as they are small, light and can conduct electricity. They can also be used in plastics to increase the strength of the plastic without adding much mass/ weight. For example, some sports equipment (like tennis rackets) are being made from plastics with nanoparticles in them.
Fullerenes can be used to cage other molecules; the fullerenes go around the outside of another molecule. This is useful in medicine as we can trap a drug inside a fullerene and use the fullerene to deliver the drug to a specific part of the body. For example, nanoparticles have the potential to treat cancer by delivering chemotherapy drugs directly to the cancer cells. This will reduce the number of healthy body cells being destroyed by chemotherapy. Nanoparticles are currently being used in wound dressing. Manufacturers add silver nanoparticles to the fibres in the wound dressing and this gives the wound dressing antibacterial properties. The uses of fullerenes in medicine is currently being researched.
Fullerenes also have a very high surface area. We can attach a catalyst for a reaction to a fullerene, which makes the catalyst more effective (a catalyst is something that speed up the rate of a reaction without being used up in the reaction).
Fullerenes can be made into nanotubes that are very long in comparison with their diameter. The nanotubes have delocalised electrons, which means that they can conduct electricity and heat. They also have very high tensile strength, which means that they do not break when they are stretched. Nanotubes can be used in electronics as they are small, light and can conduct electricity. They can also be used in plastics to increase the strength of the plastic without adding much mass/ weight. For example, some sports equipment (like tennis rackets) are being made from plastics with nanoparticles in them.
Silicon Dioxide
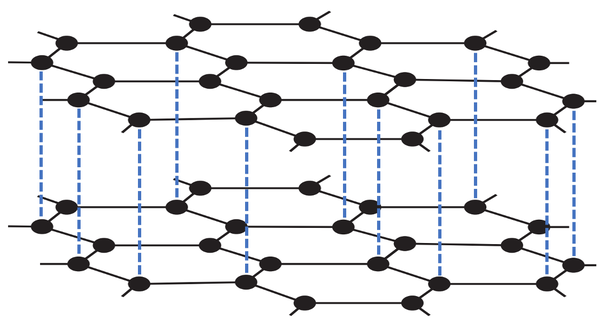
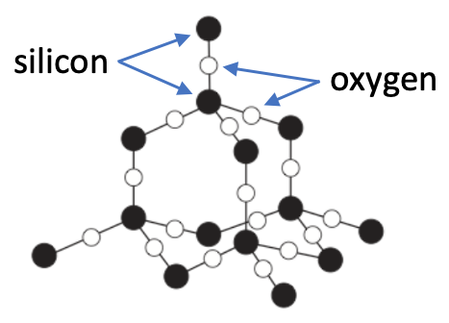
The final exam of a giant covalent structure is silicon dioxide, which is made from silicon and oxygen. Silicon dioxide is sometimes referred to as silica. Silicon dioxide is found in sand. There is a diagram of silicon dioxide below.
The final exam of a giant covalent structure is silicon dioxide, which is made from silicon and oxygen. Silicon dioxide is sometimes referred to as silica. Silicon dioxide is found in sand. There is a diagram of silicon dioxide below.