Back to C2 Home
C2 H) Metallic Bonding
C2 H) Metallic Bonding
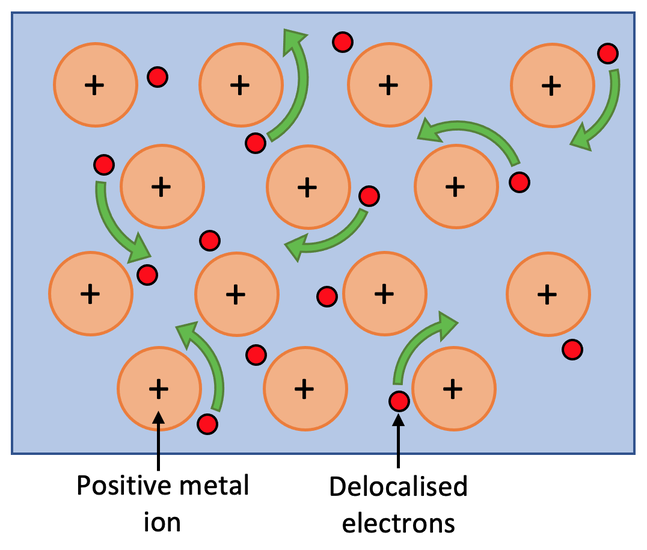
The atoms in metals are held together by metallic bonds. Metals also have delocalised electrons, which are electrons that are able to freely move throughout the whole metal. The delocalised electrons leave behind positive metal ions. The attraction between the positive metal ions and the delocalised electrons is metallic bonding. Metallic bonding is also referred to as “positive ions in a sea of delocalised negative electrons”. There is a diagram of metallic bonding below and on the diagram, you can see the positive ions and the delocalised electrons.
Metallic bonds are found in both pure metallic elements and alloys (an alloy is a mixture of two or more elements where one element is a metal).
Here are some of the characteristics of substances that have metallic bonds:
- The forces of attraction between the positive metal ions and the delocalised negative electrons are very strong. This means that a lot of energy is required to break these bonds, which is why the melting and boiling points of metals are quite high. Metals are usually solid at room temperature.
- Metallic bonding involves delocalised electrons. These delocalised electrons can carry a current and thermal energy (heat), which is why metals are good conductors of electricity and heat.
- Most metals are malleable, which means that they can be hammered or pressed into shapes without breaking. This is because the layers in the metal can slide over one another.
Alloys
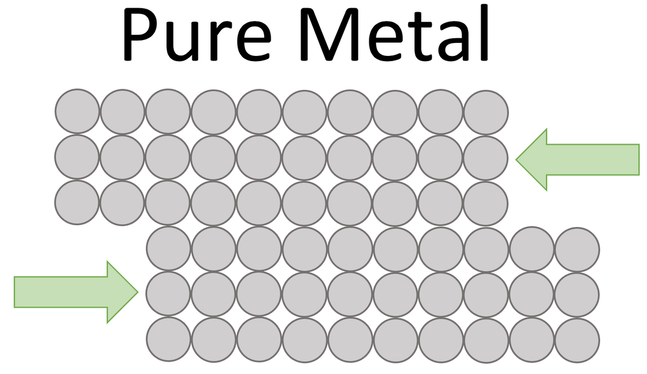
The atoms in a pure metal have a regular arrangement due to the metallic bonding. In this regular structure, the layers can easily slide over one another. This means that pure metals are usually too soft for certain uses. The arrangement of the atoms in a pure metal is shown below.
The atoms in a pure metal have a regular arrangement due to the metallic bonding. In this regular structure, the layers can easily slide over one another. This means that pure metals are usually too soft for certain uses. The arrangement of the atoms in a pure metal is shown below.
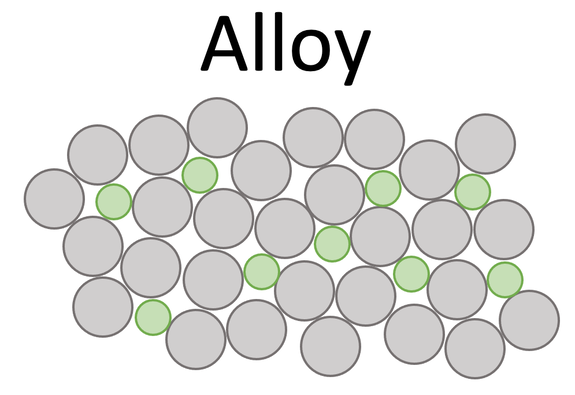
We can increase the strength of a metal by making it into an alloy. An alloy is a mixture of two or more elements where one of the elements is a metal. The other elements that we add into the alloy will be a different size to the original metal. These different sized elements will disrupt the regular arrangement of the original metal atoms. This results in it being harder for the layers to slide over one another, which results in the alloy being stronger than the pure metal. The arrangement of the atoms in an alloy is shown below.
By comparing the structure of the pure metal and the alloy, we can see that the pure metal has regular layers that can easily slide over one another meaning that the pure metal is not that strong. Whereas, for the alloy, the other elements disrupt the layers, which means that it is harder for the layers to slide over one another which results in the alloy being stronger than the pure metal.
We do not need to add large quantities of another element to a metal to increase its strength. For example, steel is an alloy of iron and carbon. Steel is used for building skyscrapers, car bodies and fences. The concentration of carbon in steel for all of these uses is between 0.05-1% (the vast majority of the alloy is iron).