Back to C2 Home
C2 I) States of Matter
C2 I) States of Matter
We can use Particle Theory to explain the properties of the 3 different states of matter; solids, liquids and gases. The particles in each of these states have different amounts of energy. The particles are the same particles in each of the 3 states. For example, water is still H2O if it is ice (solid), water (liquid) or water vapour (gas). We can use Particle Theory to explain how the particles behave in the three different states. The particles in the particle theory could be atoms, molecules or ions.
Solids
There are strong forces of attraction between the particles in a solid, which results in the particles forming a regular lattice arrangement. Solids have a fixed shape and volume and they cannot flow. This is because the particles remain in their fixed positions in the regular lattice structure. If I was to place an ice cube (solid water) into an empty glass, the ice cube will keep its shape and not fill the container. As the temperature of a solid increases, the particles have more energy which results in them vibrating more around their fixed positions. The arrangement of the particles in a solid looks like what is shown below.
There are strong forces of attraction between the particles in a solid, which results in the particles forming a regular lattice arrangement. Solids have a fixed shape and volume and they cannot flow. This is because the particles remain in their fixed positions in the regular lattice structure. If I was to place an ice cube (solid water) into an empty glass, the ice cube will keep its shape and not fill the container. As the temperature of a solid increases, the particles have more energy which results in them vibrating more around their fixed positions. The arrangement of the particles in a solid looks like what is shown below.
Liquids
There are weaker forces of attraction between the particles in a liquid compared to when the particles are in a solid. The particles are close together in a liquid. But the particles can move past each other and flow, which means that a liquid will take the shape of the container that they are put in. If I was to pour some liquid water into a cup, the water would fill the shape of the cup. Liquids have a fixed volume – they cannot be compressed or squashed. The particles in liquids have more energy than in solids. As the temperature of the liquid increases, the particles have more energy, which results in the particles moving around faster. The arrangement of the particles in a liquid looks like what is shown below.
There are weaker forces of attraction between the particles in a liquid compared to when the particles are in a solid. The particles are close together in a liquid. But the particles can move past each other and flow, which means that a liquid will take the shape of the container that they are put in. If I was to pour some liquid water into a cup, the water would fill the shape of the cup. Liquids have a fixed volume – they cannot be compressed or squashed. The particles in liquids have more energy than in solids. As the temperature of the liquid increases, the particles have more energy, which results in the particles moving around faster. The arrangement of the particles in a liquid looks like what is shown below.
Gases
Out of the 3 states, gases have the weakest force of attraction between the particles. The particles in a gas are far apart and are in a random arrangement. The particles move quickly in random directions and at a range of speeds. A gas will completely fill the container that they are placed in, and they can be compressed or squashed. This means that gases do not have a definite shape or volume. Out of the 3 states, the particles in a gas have the most energy. As the temperature of a gas increases, the particles will have more energy, which causes them to move around faster. This causes the pressure of the gas to increase.
Out of the 3 states, gases have the weakest force of attraction between the particles. The particles in a gas are far apart and are in a random arrangement. The particles move quickly in random directions and at a range of speeds. A gas will completely fill the container that they are placed in, and they can be compressed or squashed. This means that gases do not have a definite shape or volume. Out of the 3 states, the particles in a gas have the most energy. As the temperature of a gas increases, the particles will have more energy, which causes them to move around faster. This causes the pressure of the gas to increase.
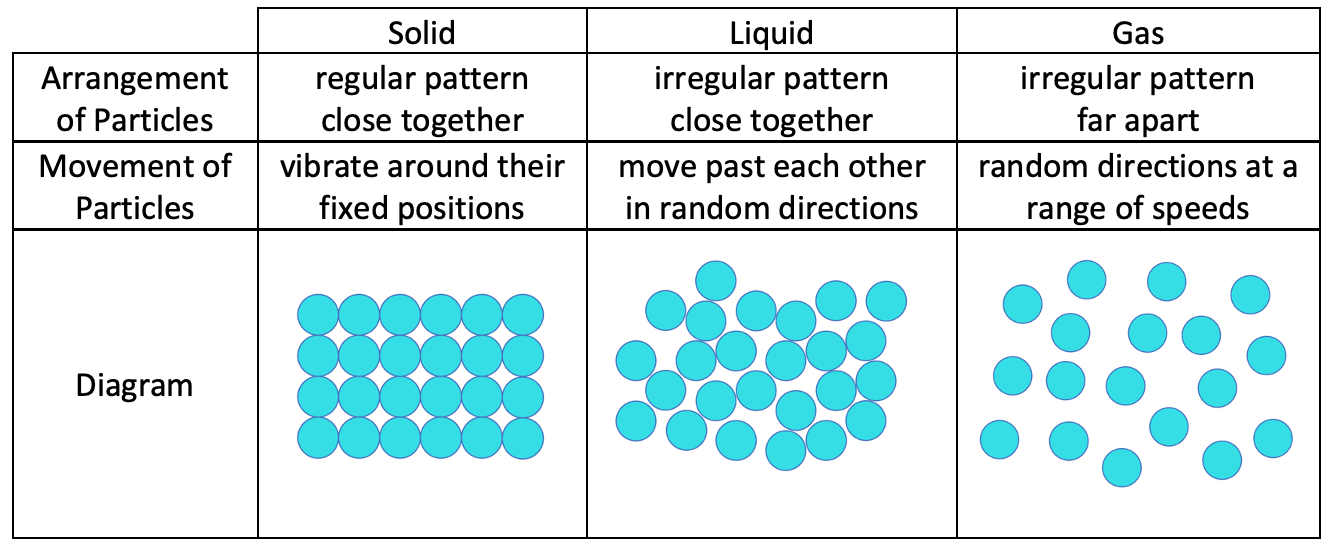
Comparison
Here is a comparison of the characteristics of the particles in the 3 different states.
A Few Drawbacks of the Model
The particle theory model is a good model for explaining the characteristics of the particles in the 3 different states. However, it does have a few drawbacks. One drawback is that the particles in real life are not solid spheres; instead they are atoms, molecules and ions. Also, the particle theory model does not show how strong the forces are between each of the particles in the model; it does not explain why the forces are stronger for certain substances (like giant ionic compounds, such as sodium chloride) and weaker for other substances (like simple molecular substances, such as water).
The particle theory model is a good model for explaining the characteristics of the particles in the 3 different states. However, it does have a few drawbacks. One drawback is that the particles in real life are not solid spheres; instead they are atoms, molecules and ions. Also, the particle theory model does not show how strong the forces are between each of the particles in the model; it does not explain why the forces are stronger for certain substances (like giant ionic compounds, such as sodium chloride) and weaker for other substances (like simple molecular substances, such as water).
State Symbols
In chemical equations, you will sometimes see state symbols written in the subscript next to the different substances involved in the reaction. There are 4 state symbols and they are:
We are going to use the reaction of zinc and sulfuric acid to produce zinc sulfate and hydrogen. The reaction equation is shown below:
In chemical equations, you will sometimes see state symbols written in the subscript next to the different substances involved in the reaction. There are 4 state symbols and they are:
- Solid – (s)
- Liquid – (l)
- Gas – (g)
- Aqueous – (aq) – means ‘dissolved in water’
We are going to use the reaction of zinc and sulfuric acid to produce zinc sulfate and hydrogen. The reaction equation is shown below:
From the state symbols, we can see that zinc is a solid, sulfuric acid and zinc sulfate are aqueous (dissolved in water), and hydrogen is a gas.