Back to C2 Home
C2 J) Changing State
C2 J) Changing State
Changes of state occur when the amount of energy that the particles have increases. A change in state does not change what the particles are, it just changes the arrangement of those particles; the changing of state is a physical change and not a chemical change (a chemical change is where the substance/ particles change as a result of a chemical reaction). For example, water is still H2O if it is ice (solid), water (liquid) or water vapour (gas). I am now going to go through how the energy of the particles causes the states to change.
Solid to Liquid to Gas
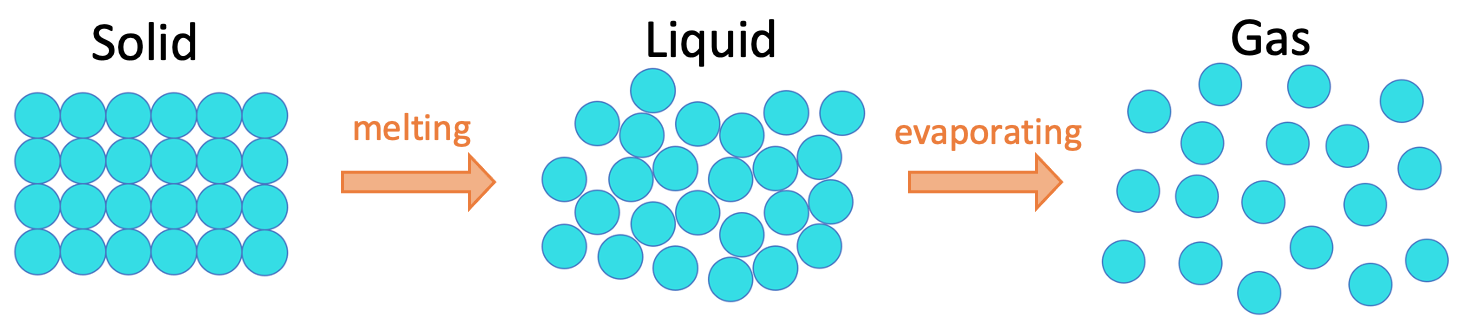
We are now going to look at what happens to the particles and the states as we heat up a solid.
When the particles in a solid are heated, they gain more energy and start to vibrate more around their fixed positions. The increase in vibration of the particles weakens the forces that hold the solid together.
Eventually at a certain temperature (the melting point), the particles will have enough energy to break free from their fixed positions in a solid and become a liquid. The change in state from a solid to a liquid is known as melting.
If we continue to heat a liquid, the particles gain more energy, which weakens the forces holding the liquid together. At a certain temperature (known as the boiling point), the particles will have enough energy to break free of their positions in the liquid and the liquid will turn into a gas. The change in state from a liquid to a gas is known as evaporating.
We are now going to look at what happens to the particles and the states as we heat up a solid.
When the particles in a solid are heated, they gain more energy and start to vibrate more around their fixed positions. The increase in vibration of the particles weakens the forces that hold the solid together.
Eventually at a certain temperature (the melting point), the particles will have enough energy to break free from their fixed positions in a solid and become a liquid. The change in state from a solid to a liquid is known as melting.
If we continue to heat a liquid, the particles gain more energy, which weakens the forces holding the liquid together. At a certain temperature (known as the boiling point), the particles will have enough energy to break free of their positions in the liquid and the liquid will turn into a gas. The change in state from a liquid to a gas is known as evaporating.
Gas to Liquid to Solid
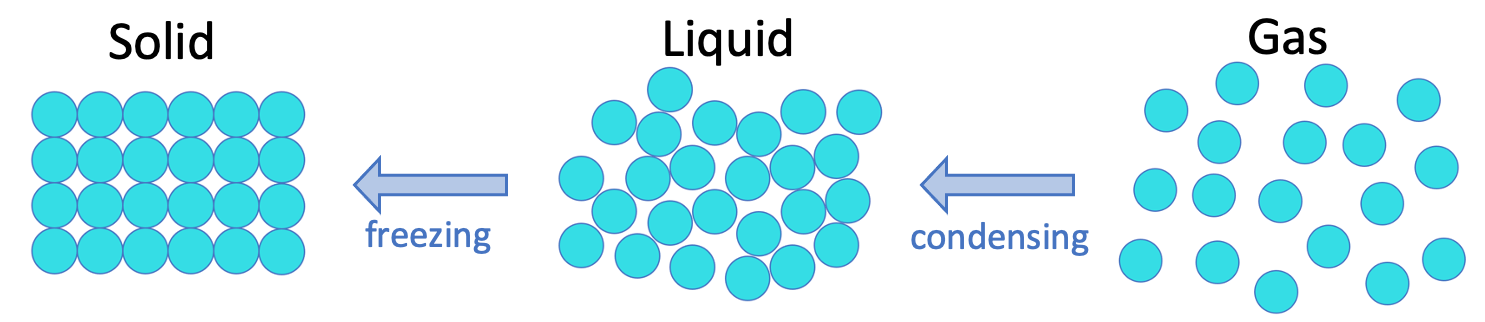
We are now going to look at what happens to the particles and the states as we cool a gas; essentially the opposite happens.
As a gas cools, the particles lose some of their energy. This means that they do not have enough energy to overcome the forces of attraction between the particles, which results in bonds starting to form between the particles. At the boiling point, so many bonds have formed between the particles, which results in the gas becoming a liquid. The changing in state from a gas to a liquid is known as condensing.
If we continue to cool a liquid, the particles will have less energy. This will result in bonds forming between the particles as the particles do not posses enough energy to overcome the attraction between the particles. At the melting point, so many bonds have been formed between the particles, which results in the liquid becoming a solid. The changing in state from a liquid to a solid is known as freezing.
We are now going to look at what happens to the particles and the states as we cool a gas; essentially the opposite happens.
As a gas cools, the particles lose some of their energy. This means that they do not have enough energy to overcome the forces of attraction between the particles, which results in bonds starting to form between the particles. At the boiling point, so many bonds have formed between the particles, which results in the gas becoming a liquid. The changing in state from a gas to a liquid is known as condensing.
If we continue to cool a liquid, the particles will have less energy. This will result in bonds forming between the particles as the particles do not posses enough energy to overcome the attraction between the particles. At the melting point, so many bonds have been formed between the particles, which results in the liquid becoming a solid. The changing in state from a liquid to a solid is known as freezing.
The melting and boiling points are different for different substances. This is because the melting and boiling points are dependent upon the forces of attraction between the particles. The stronger the forces of attraction between the particles, the higher the melting and boiling points are. This is because more energy (a greater temperature) is required in order to overcome these stronger forces of attraction between the particles, hence why the melting and boiling points are higher.
Summary of all Changes of State
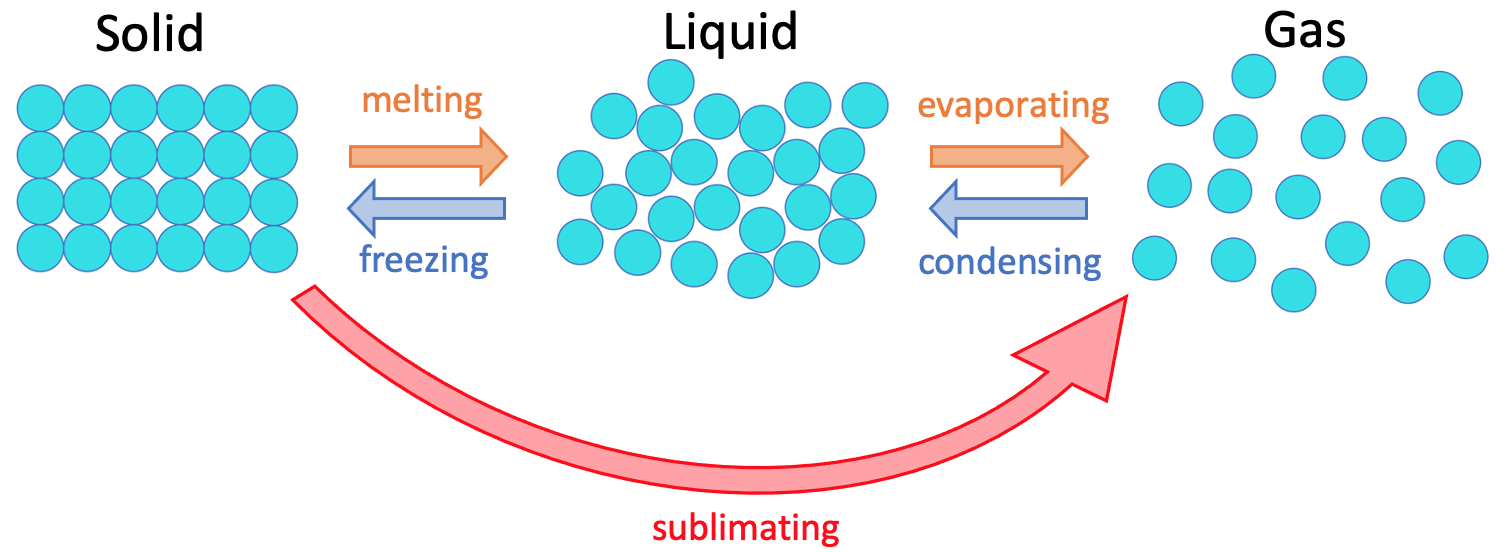
There is one more change of state that we are going to look at. This change in state is called sublimating/ sublimation, and it is where a solid turns straight into a gas. The 5 changes in state are shown below.
There is one more change of state that we are going to look at. This change in state is called sublimating/ sublimation, and it is where a solid turns straight into a gas. The 5 changes in state are shown below.
Predicting States
We may be given the melting and boiling points for a few substances and be asked to find what state a substance is at a certain temperature. These questions can be a little bit tricky because they often involve negative numbers. Therefore, the easiest way to answer questions like this is to draw a number line. We then mark on the melting and boiling points, and the temperature that we asked to make the prediction about the state for.
Example 1
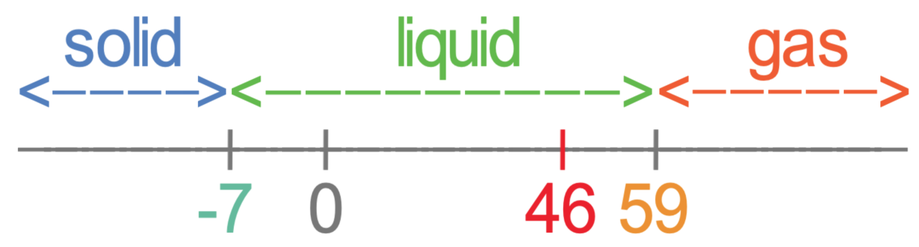
Bromine has a melting point of -7°C and a boiling point of 59°C. What state will bromine be at 46°C?
The first step in answering this question is to draw a number line with the melting point (-7°C) and the boiling point (59°C) on. We also put 0 on the number line.
We may be given the melting and boiling points for a few substances and be asked to find what state a substance is at a certain temperature. These questions can be a little bit tricky because they often involve negative numbers. Therefore, the easiest way to answer questions like this is to draw a number line. We then mark on the melting and boiling points, and the temperature that we asked to make the prediction about the state for.
Example 1
Bromine has a melting point of -7°C and a boiling point of 59°C. What state will bromine be at 46°C?
The first step in answering this question is to draw a number line with the melting point (-7°C) and the boiling point (59°C) on. We also put 0 on the number line.
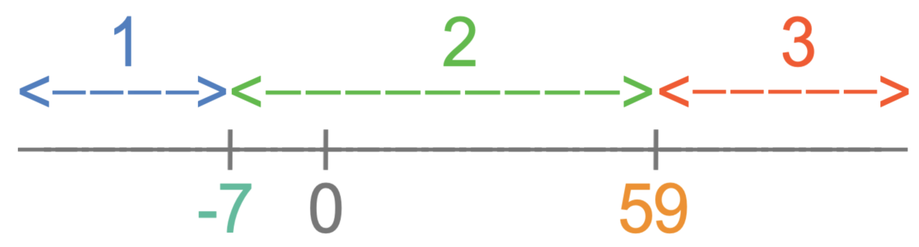
The number line is useful as it allows us to quickly see what state certain temperatures will be. There are 3 different parts on the number line, which I have labelled as 1, 2 and 3. The different parts are:
The next step is to mark on the temperature that we are looking for the state of. The question is asking us to find the state of bromine at 46°C, so we mark 46°C onto the number line.
- Part 1 is temperatures that are less than the melting point, which is -7°C for bromine. At temperatures less than the melting point, the substance will be a solid.
- Part 2 is temperatures that are greater than the melting point and less than the boiling point, which are temperatures between -7°C and 59°C. At temperatures in between the melting point and boiling point, the substance will be a liquid.
- Part 3 is temperatures above the boiling point, which is 59°C for bromine. At temperatures above the boiling point, the substance will be a gas.
The next step is to mark on the temperature that we are looking for the state of. The question is asking us to find the state of bromine at 46°C, so we mark 46°C onto the number line.
By marking 46°C on the number line, we can see that bromine will be a liquid at 46°C. This is because 46°C is greater than the melting point (-7°C) and less than the boiling point (59°C) – it is in section 2.
Example 2
Chlorine has a melting point of -101°C and a boiling point of -34°C. What state will chlorine be at -125°C?
The first step in answering this question is to draw a number line with the melting point (-101°C) and the boiling point (-34°C) on.
Chlorine has a melting point of -101°C and a boiling point of -34°C. What state will chlorine be at -125°C?
The first step in answering this question is to draw a number line with the melting point (-101°C) and the boiling point (-34°C) on.
We now mark on the temperature that we are looking for the state of, which is -125°C.
By marking -125°C on the number line, we can see that chlorine will be a solid at -125°C. This is because -125°C is less than the melting point (-101°C); it is in section 1.