Back to C2 Home
C2: Quiz 10 – Answers
C2: Quiz 10 – Answers
1) Physical change as no chemical reaction has taken place; the particles are still water (H2O) when ice (solid) becomes water (liquid)
2)
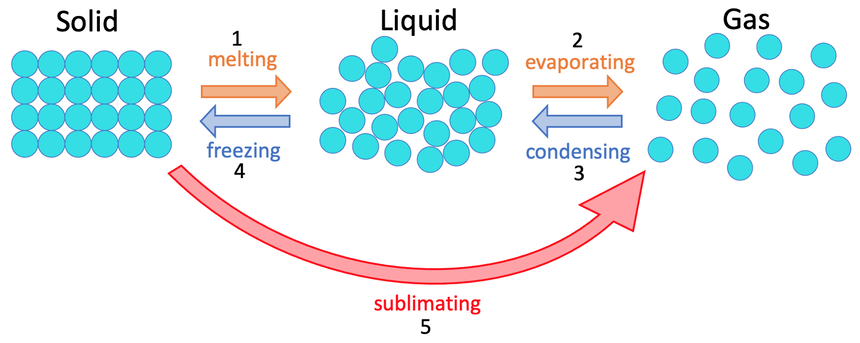
1 – melting
2 – evaporating or boiling
3 – condensing
4 – freezing or solidifying
5 – sublimating
2)
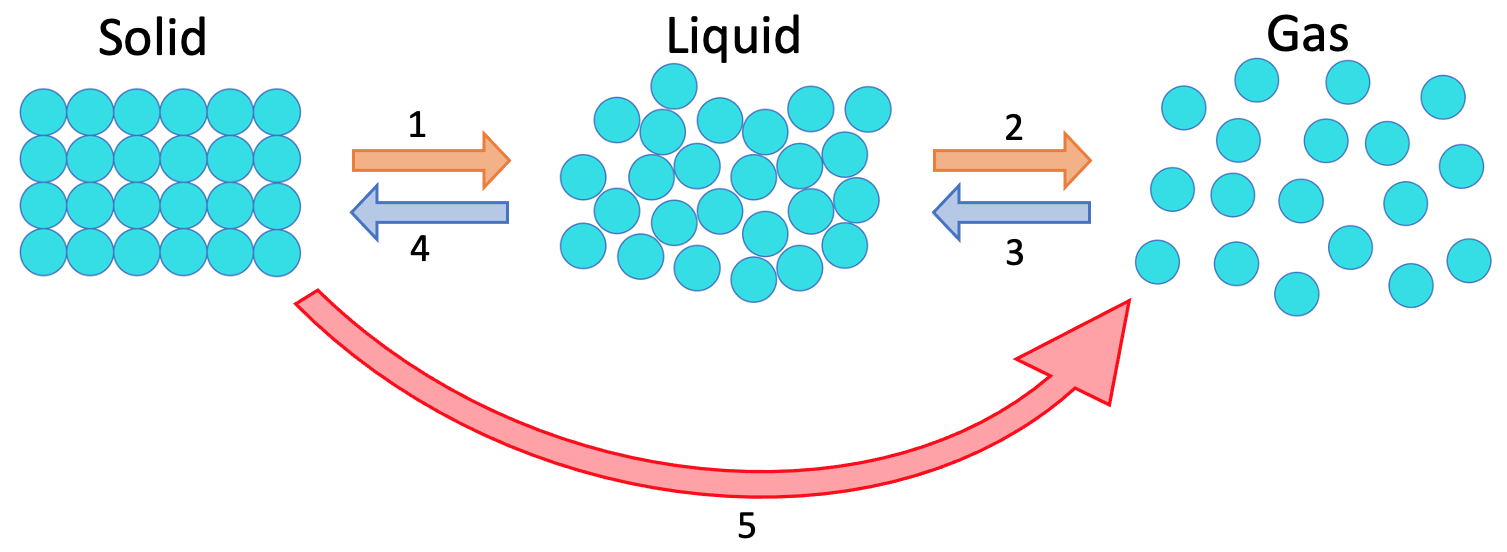
1 – melting
2 – evaporating or boiling
3 – condensing
4 – freezing or solidifying
5 – sublimating
3) The particles in a solid gain more energy which causes them to vibrate more around their fixed positions. The increase in vibration/ energy of the particles weakens the forces that hold the solid together. Eventually at a certain temperature (the melting point), the particles have enough energy to break free from their fixed positions in the solid and become a liquid
4) As a gas cools, the particles lose some of their energy. This means that they do not have enough energy to overcome the forces of attraction between the particles, which results in bonds starting to form between the particles. At the boiling point, so many bonds have formed between the particles, which results in the gas becoming a liquid.
5)
a) Liquid
b) Gas
c) Solid
6)
a) Solid
b) Gas
c) Solid
d) Liquid
7)
a) Gas
b) Liquid
c) Gas
d) Solid
8)
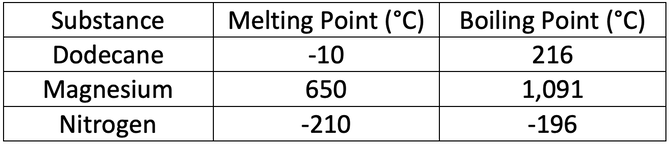
a) Magnesium
b) Nitrogen
c) Dodecane
d) Dodecane, magnesium & nitrogen
4) As a gas cools, the particles lose some of their energy. This means that they do not have enough energy to overcome the forces of attraction between the particles, which results in bonds starting to form between the particles. At the boiling point, so many bonds have formed between the particles, which results in the gas becoming a liquid.
5)
a) Liquid
b) Gas
c) Solid
6)
a) Solid
b) Gas
c) Solid
d) Liquid
7)
a) Gas
b) Liquid
c) Gas
d) Solid
8)
a) Magnesium
b) Nitrogen
c) Dodecane
d) Dodecane, magnesium & nitrogen
Questions
1) If H2O was to change state from solid (ice) to liquid (water), would that be a physical or chemical change? Explain your answer.
2) The diagram below shows the three different states of matter.
What are the names for the 5 different changes of state on the diagram above?
3) Explain how a substance changes state from a solid to a liquid.
4) Explain how a substance changes state from a gas to a liquid.
5) Ethanol has a melting point of -114°C and a boiling point of 78°C. Predict the states for ethanol at the following temperatures:
a) 10°C
b) 178°C
c) -120°C
6) Sodium chloride has a melting point of 801°C and a boiling point of 1,465°C. Predict the states for sodium chloride at the following temperatures:
a) 175°C
b) 2500°C
c) -120°C
d) 1,200°C
7) Fluorine has a melting point of -220°C and a boiling point of -188°C. Predict the states for fluorine at the following temperatures:
a) 160°C
b) -200°C
c) -67°C
d) -251°C
8) The table below shows the melting and boiling points of three different substances.
3) Explain how a substance changes state from a solid to a liquid.
4) Explain how a substance changes state from a gas to a liquid.
5) Ethanol has a melting point of -114°C and a boiling point of 78°C. Predict the states for ethanol at the following temperatures:
a) 10°C
b) 178°C
c) -120°C
6) Sodium chloride has a melting point of 801°C and a boiling point of 1,465°C. Predict the states for sodium chloride at the following temperatures:
a) 175°C
b) 2500°C
c) -120°C
d) 1,200°C
7) Fluorine has a melting point of -220°C and a boiling point of -188°C. Predict the states for fluorine at the following temperatures:
a) 160°C
b) -200°C
c) -67°C
d) -251°C
8) The table below shows the melting and boiling points of three different substances.
You may get more than one substance for each of the following questions.
a) Which of the substances will be a solid at 0°C
b) Which of the substances will be a gas at -150°C
c) Which of the substances will be a liquid at 85°C
d) Which of the substances will be a gas at 1,200°C
a) Which of the substances will be a solid at 0°C
b) Which of the substances will be a gas at -150°C
c) Which of the substances will be a liquid at 85°C
d) Which of the substances will be a gas at 1,200°C