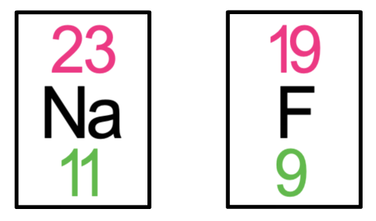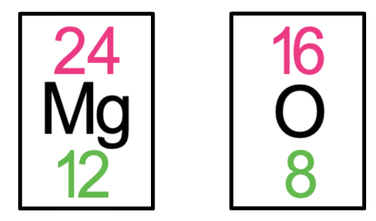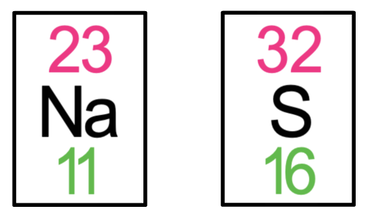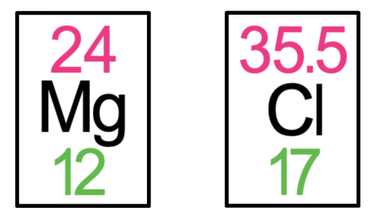C2: Quiz 1 – Answers
2) Electrostatic attraction/ electrostatic force
3)
a)
4)
a)
5)
a)
6)
a)
7)
Calcium loses 2 electrons and oxygen gains 2 electrons
2 electrons are transferred
Calcium has a 2+ charge
Oxide has a 2- charge
Click here for a printable PDF of the diagrams in this quiz. Also, some of the diagrams may have empty electron shells – do not let this put you off when you are answering these questions.
1) What is ionic bonding?
2) What is the name of the force that holds ionic compounds together?
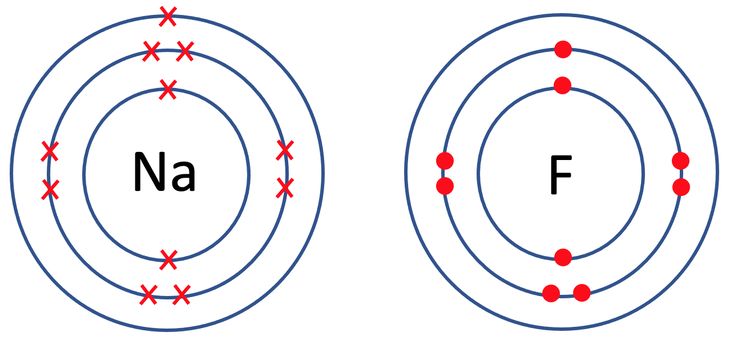
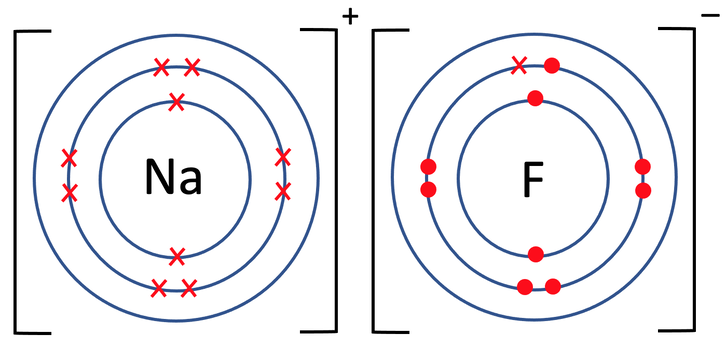
3) Sodium fluoride (NaF) is an ionic compound that is made out of 1 sodium atom and 1 fluorine atom. The tiles in the periodic table for sodium and fluorine are shown below.
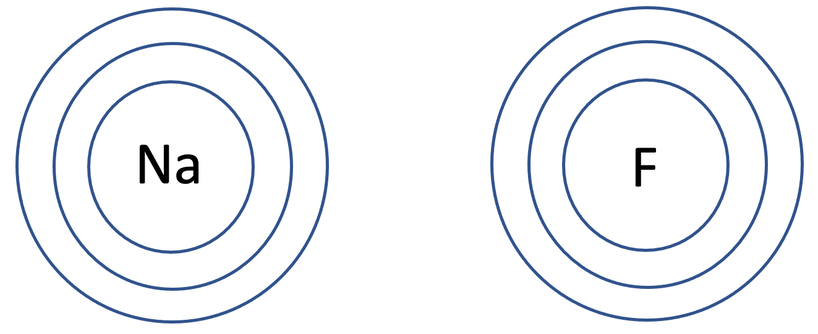
a) Draw the electronic configuration of sodium and fluorine prior to them bonding.
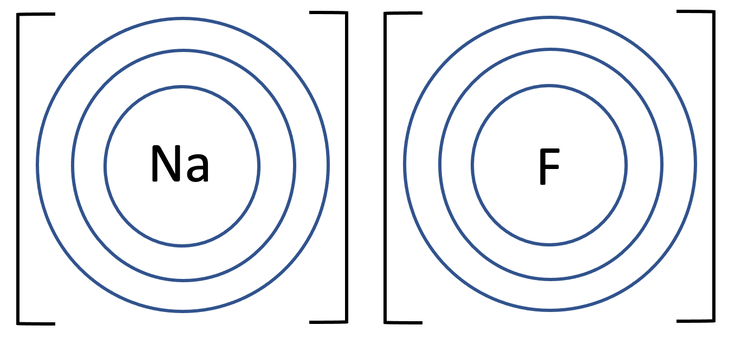
b) Draw the electronic configuration after the sodium and fluorine have bonded. Remember to include your charges on the outside of the square brackets.
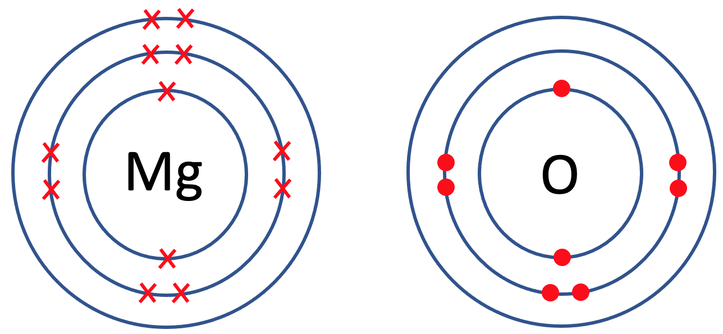
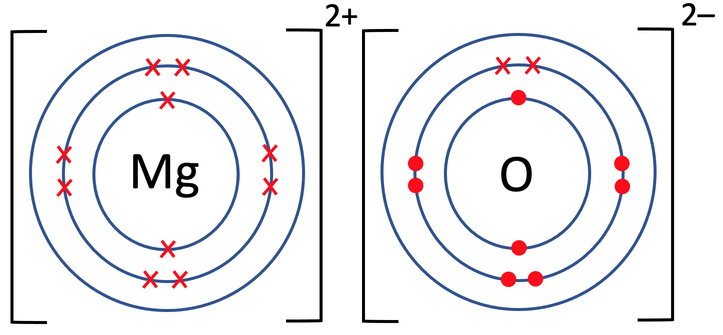
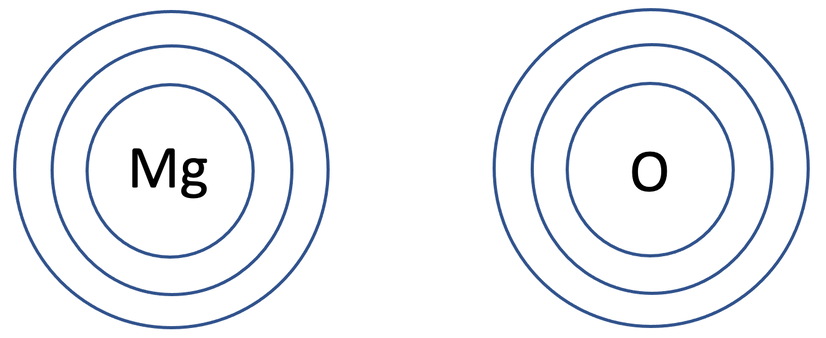
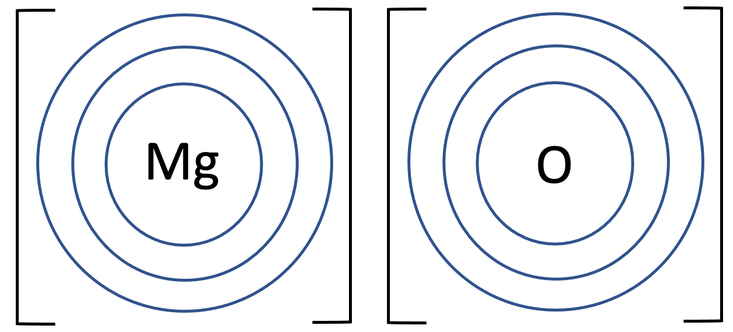
4) Magnesium oxide (MgO) is an ionic compound that is made out of 1 magnesium atom and 1 oxygen atom. The tiles in the periodic table for magnesium and oxygen are shown below.
a) Draw the electronic configuration of magnesium and oxygen prior to them bonding.
b) Draw the electronic configuration after the magnesium and oxygen have bonded. Remember to include your charges on the outside of the square brackets.
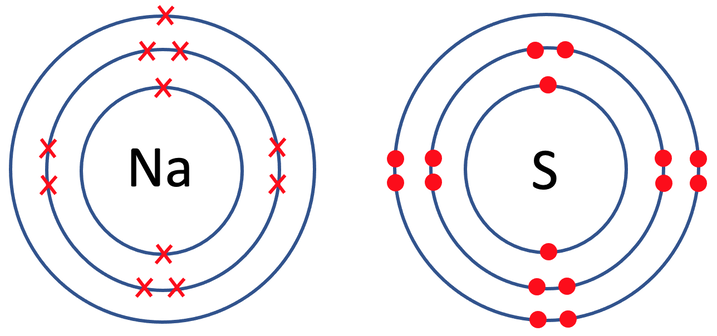
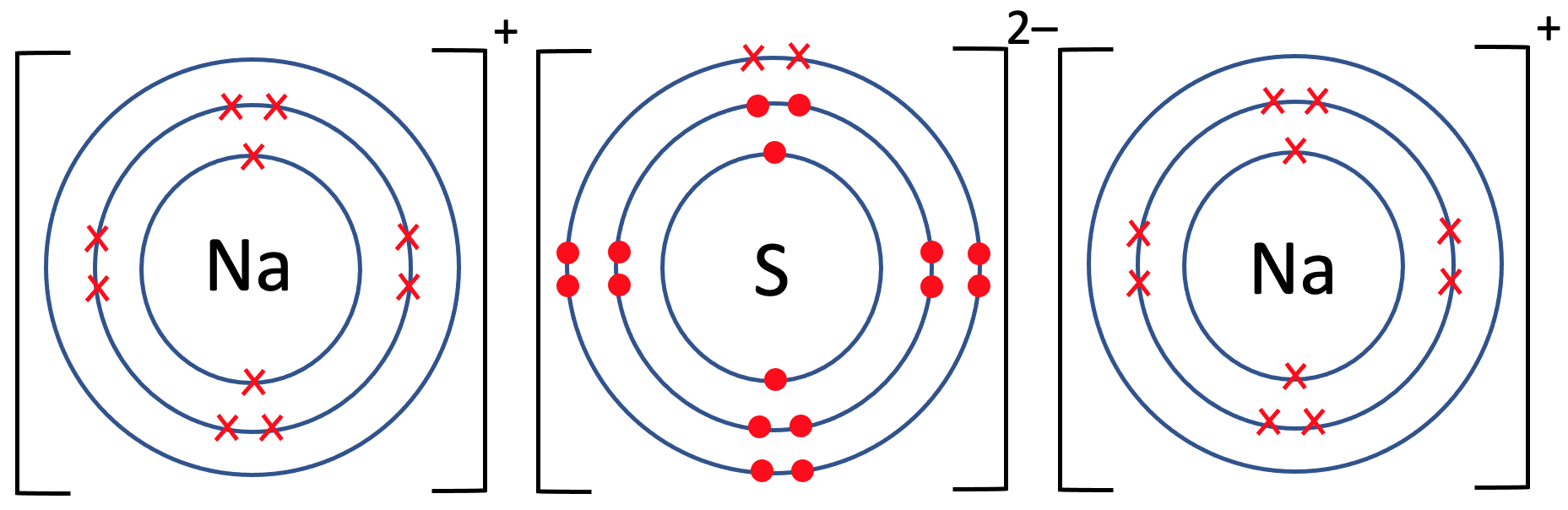
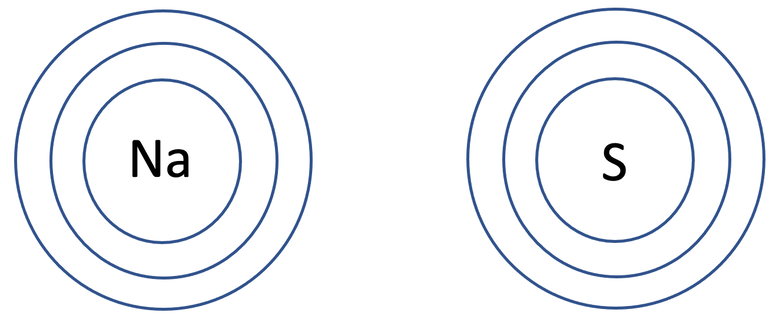
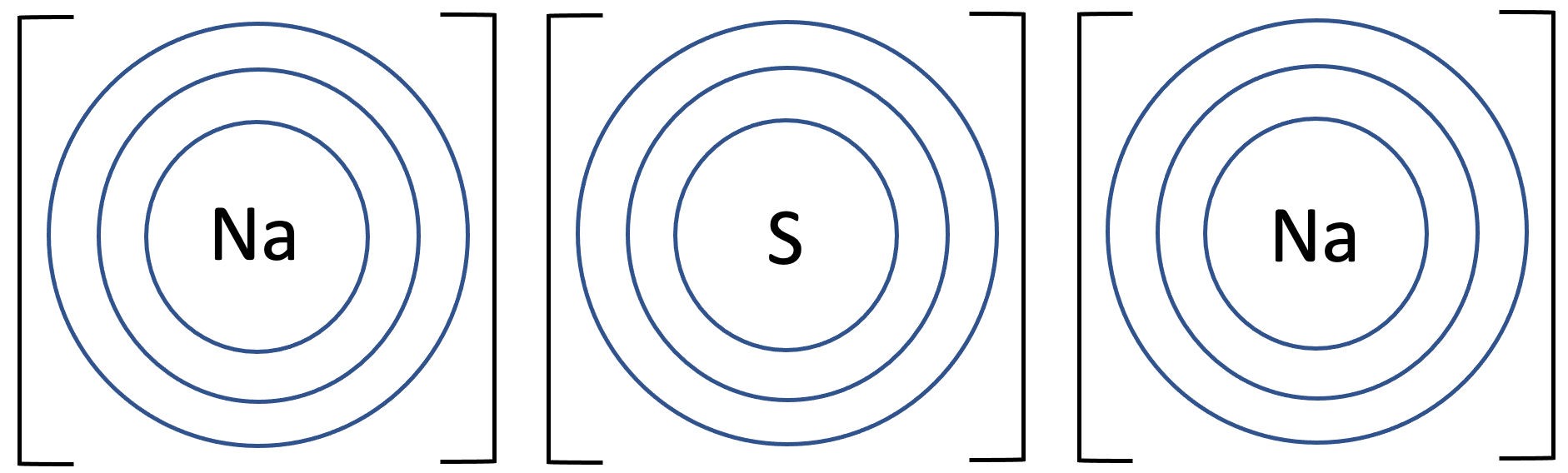
5) Sodium sulphide has the formula Na2S. The tiles in the periodic table for sodium and sulfur are shown below.
a) Draw the electronic configuration of a sodium and sulfur atom prior to them bonding.
b) Draw the electronic configuration after the sodium and sulfur atoms have bonded. Remember to include your charges on the outside of the square brackets.
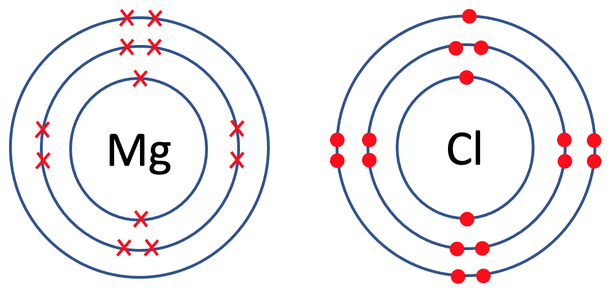
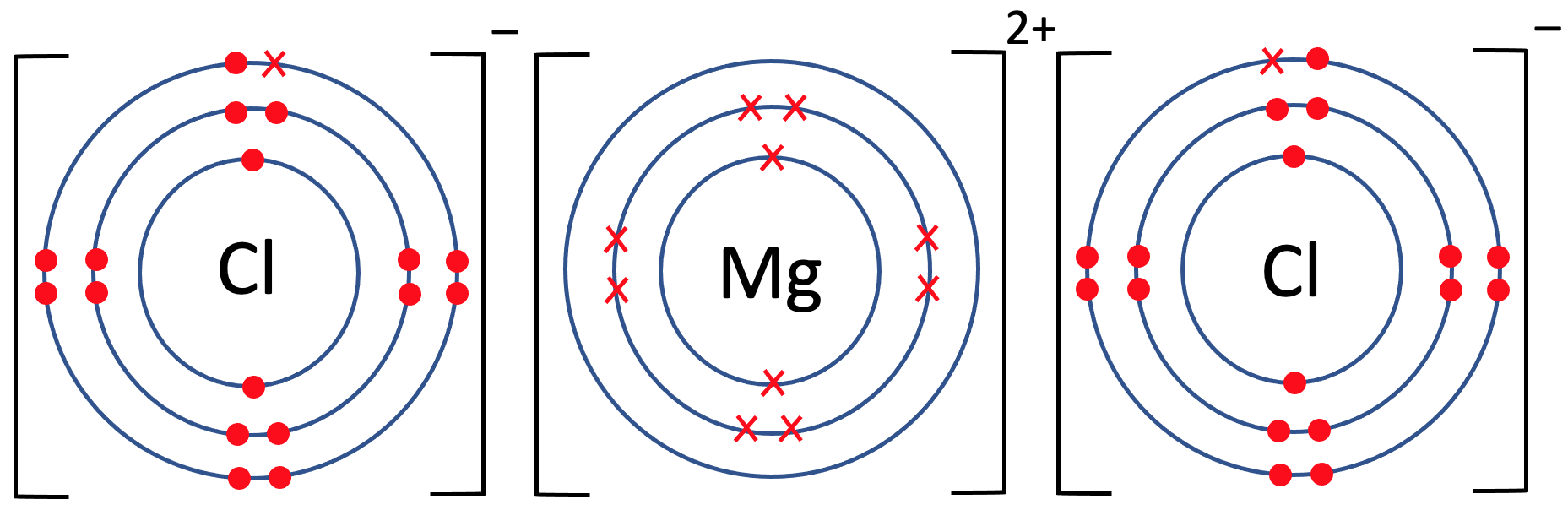
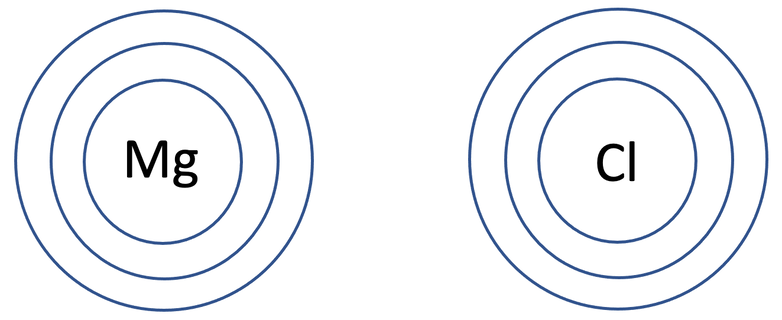
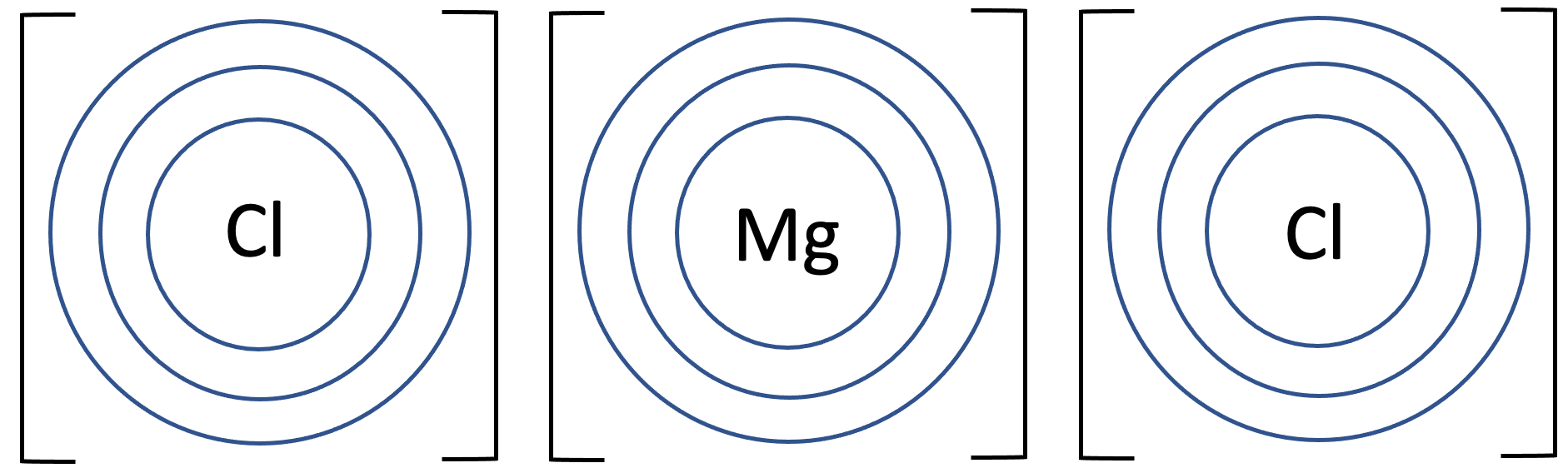
6) Magnesium chloride has the formula MgCl2. The tiles in the periodic table for magnesium and chlorine are shown below.
a) Draw the electronic configuration of a magnesium and chlorine atom prior to them bonding.
b) Draw the electronic configuration of magnesium chloride. Remember to include your charges on the outside of the square brackets.
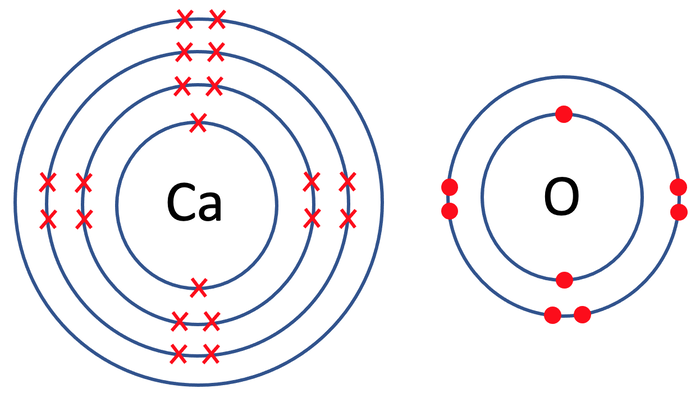
7) This final question is a past paper question.
The diagram below shows the electronic structure of an oxygen and calcium atom.
Describe how the calcium atom and oxygen atom form calcium oxide. You should give the charges on each of the ions formed and discuss what happens to the electrons. [4 marks]