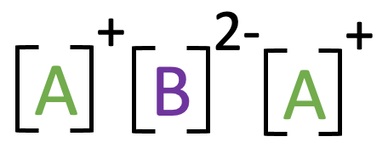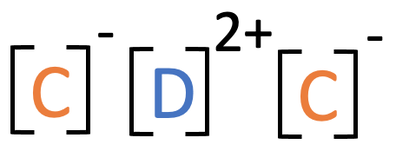Back to C2 Home
C2: Quiz 2 – Answers
C2: Quiz 2 – Answers
1) Give, receive, positive, negative
Metals want to give electrons and non-metals want to receive electrons. This results in the metals becoming positive ions, and the non-metals becoming negative ions.
2)
a) 1
b) Lose 1 electron
c) + or 1+
d) Na –> Na+ + e-
3)
a) Lose 2 electrons
b) Ca –> Ca2+ + 2 e-
4)
a) Gain 2 electrons
b) S + 2 e- –> S2-
5) Cl + e- –> Cl-
6)
a) Metal
b) Al –> Al3+ + 3 e-
7)
a) Group 1 because the ion of A has a charge of 1+/ it has lost 1 electron
b) Group 6 because the ion of B has a charge of 2-/ it has gained 2 electrons
8)
a) Group 7 because the ion of C has a charge of 1-/ it has gained 1 electron
b) Group 2 because the ion of D has a charge of 2+/ it has lost 2 electrons
Questions
1) Use the words “positive”, “negative”, “give” and “receive” to fill in the blanks in the sentence below.
Metals want to __________ electrons and non-metals want to __________ electrons. This results in the metals becoming __________ ions, and the non-metals becoming __________ ions.
2)
a) How many electrons do the group 1 elements have in their outermost electron shell?
b) How many electrons do the group 1 elements want to lose?
c) What will be the charge on group 1 elements after they have lost electron(s)?
d) Sodium (Na) is a group 1 element. Write the half equation for it losing electron(s).
3)
a) Do group 2 elements want to lose or gain electrons, and how many electrons do they want to lose or gain?
b) Calcium (Ca) is a group 2 element. Write the half equation for it losing or gaining electron(s).
4) Sulfur is a group 6 element. The symbol for sulfur is S.
a) How many electrons does sulfur need to gain to have a full outer electron shell?
b) Write the half equation for sulfur gaining electron(s).
5) Write the half equation for chlorine gaining electron(s). Chorine has the symbol Cl.
Use a periodic table to help you find the group that chlorine is in – click here for a periodic table.
6) Aluminium has the symbol Al.
a) Is aluminium a metal or non-metal?
b) Write the half equation for aluminium when aluminium forms ionic bonds.
7) The ionic compound below is made out of two unknown elements; A and B.
1) Use the words “positive”, “negative”, “give” and “receive” to fill in the blanks in the sentence below.
Metals want to __________ electrons and non-metals want to __________ electrons. This results in the metals becoming __________ ions, and the non-metals becoming __________ ions.
2)
a) How many electrons do the group 1 elements have in their outermost electron shell?
b) How many electrons do the group 1 elements want to lose?
c) What will be the charge on group 1 elements after they have lost electron(s)?
d) Sodium (Na) is a group 1 element. Write the half equation for it losing electron(s).
3)
a) Do group 2 elements want to lose or gain electrons, and how many electrons do they want to lose or gain?
b) Calcium (Ca) is a group 2 element. Write the half equation for it losing or gaining electron(s).
4) Sulfur is a group 6 element. The symbol for sulfur is S.
a) How many electrons does sulfur need to gain to have a full outer electron shell?
b) Write the half equation for sulfur gaining electron(s).
5) Write the half equation for chlorine gaining electron(s). Chorine has the symbol Cl.
Use a periodic table to help you find the group that chlorine is in – click here for a periodic table.
6) Aluminium has the symbol Al.
a) Is aluminium a metal or non-metal?
b) Write the half equation for aluminium when aluminium forms ionic bonds.
7) The ionic compound below is made out of two unknown elements; A and B.
a) Suggest which group in the periodic table element A comes from and give a reason for your answer.
b) Suggest which group in the periodic table element B comes from and give a reason for your answer.
8) The ionic compound below is made out of two unknown elements; C and D
b) Suggest which group in the periodic table element B comes from and give a reason for your answer.
8) The ionic compound below is made out of two unknown elements; C and D
a) Suggest which group in the periodic table element C comes from and give a reason for your answer.
b) Suggest which group in the periodic table element D comes from and give a reason for your answer.
b) Suggest which group in the periodic table element D comes from and give a reason for your answer.