C2: Quiz 3 – Answers
1) High melting and boiling points
Because they have extremely strong electrostatic forces of attraction between the positive and negative ions which requires a lot of energy to break and change their state, thus meaning that ionic compounds have high melting and boiling points
2) Ionic compounds cannot conduct electricity in a solid. This is because they do not have any delocalised electrons (electrons that can move) and the ions are unable to move as they are in their fixed positions in the solid.
3) Aqueous means dissolved in water
4) Ionic compounds can conduct electricity when they are liquid or aqueous because the ions are able to move and can therefore carry an electric current
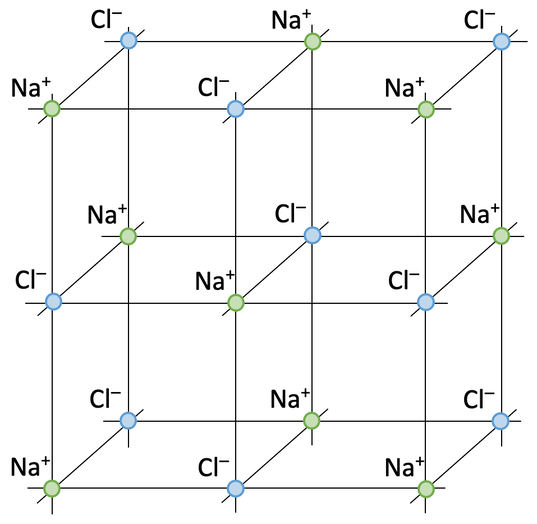
5) There are positive sodium ions (Na+) and negative fluoride ions (F-). The ions are arranged in a regular giant ionic lattice and there are very strong electrostatic forces of attraction in all directions keeping the ions in their positions
6) The ions in sodium chloride are Na+ and Cl-. The ions in magnesium oxide are Mg2+ and O2-. Therefore, there are stronger electrostatic forces of attraction in the magnesium oxide (2+ and 2-) compared to the sodium chloride (just 1+ and 1-). This means that more energy is required to overcome these stronger electrostatic forces of attraction in magnesium oxide, hence why magnesium oxide has a higher melting point than sodium chloride
7)
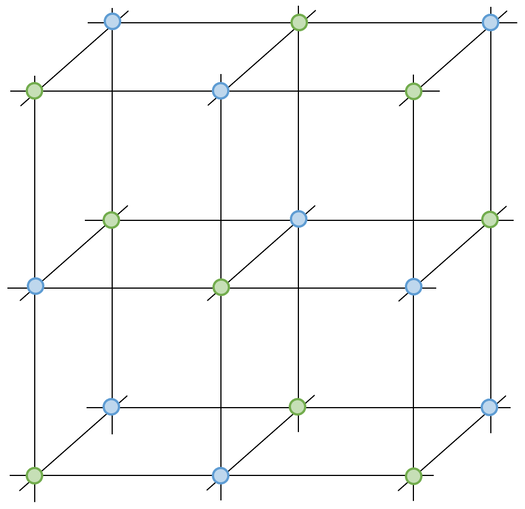
a) Make sure that all of the sodium ions are one colour and all of the chloride ions are the other colour
b) Any advantage such as:
- Shows the regular pattern of all of the positive and negative ions
- Suggests that the structure continues in all directions
- Shows that it is a 3D structure
- Implies that all of the ions are the same size
8)
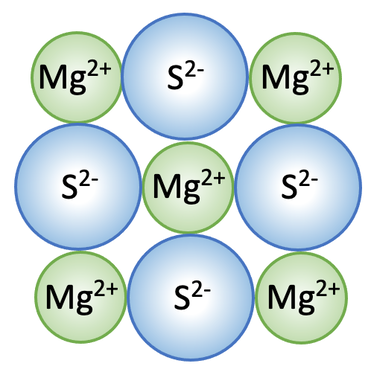
a) Mg2+ and S2-
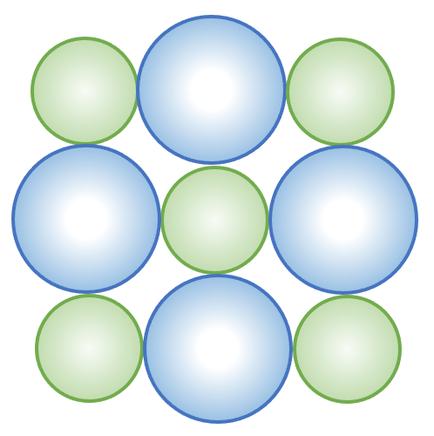
b) The magnesium ions need to be green and the sulphide ions need to be blue; this is because the magnesium ions have 2 shells and are therefore smaller than the sulphide ions that have 3 shells.
Any advantage such as:
- Shows the regular pattern of all of the positive and negative ions
- Shows the different sizes of the ions
- Doesn’t suggest that the structure continues in all directions
- Doesn’t shows that it is a 3D structure
1) Do ionic compounds have high or low melting and boiling points? Explain your answer.
2) Do ionic compounds conduct electricity in a solid state? Explain your answer.
3) What does aqueous mean?
4) Do ionic compounds conduct electricity when they are liquid or aqueous? Explain your answer.
5) A crystal of sodium fluoride (NaF) is an ionic compound. Describe the structure of a sodium fluoride crystal.
Include the ions involved, the arrangement of the ions and what holds the ions together. Do not talk about how the electrons are transferred.
6) Sodium chloride has a melting point of 801°C. Magnesium oxide has a melting point of 2,852°C. Explain why the melting point of magnesium oxide is higher than the melting point of sodium chloride.
7) Sodium chloride is made out of sodium ions (Na+) and chloride ions (Cl-).
a) Add the sodium ions and chloride ions to the diagram below.
b) Give an advantage and disadvantage of using this type of diagram to show ionic bonding.
8) Magnesium sulphide is an ionic compound; the formula for magnesium sulphide is MgS.
a) What are the two different ions involved in magnesium sulphide?
b)
i) How many electron shells does a magnesium ion in magnesium sulphide have?
ii) How many electron shells does a sulphide ion in magnesium sulphide have?
c) Complete the diagram below; be careful with the colours of the ions.
d) Give an advantage and a disadvantage of using this type of diagram to show ionic bonding.