Back to C2 Home
C2: Quiz 6
C2: Quiz 6
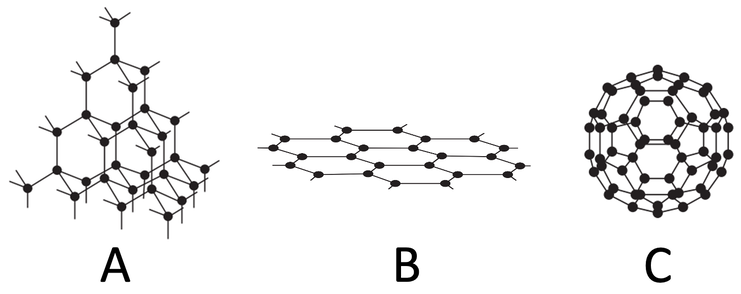
1) Name the substance for each of the diagrams below.
2) This question is all about diamond.
a) Diamond is made out of carbon. How many covalent bonds do each of the carbon atoms in diamond form?
b) Does diamond have a very high or a very low melting and boiling point? Explain your answer.
c) Does diamond conduct electricity? Explain your answer.
d) Why is diamond used in cutting tools?
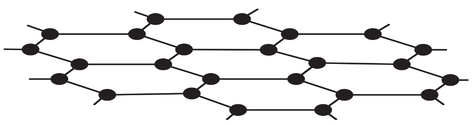
3) The diagram below is of graphene.
a) What element is graphene made from?
b) How many bonds do each of the atoms in graphene form?
c) What is the name of the shape that the atoms in graphene are arranged in?
d) Does graphene conduct electricity? Explain your answer.
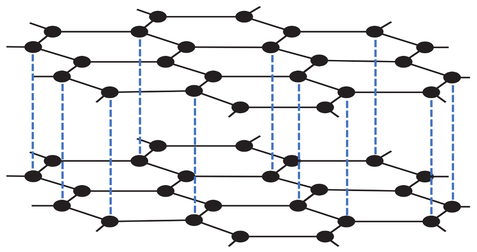
4) The diagram below shows graphite.
b) How many bonds do each of the atoms in graphene form?
c) What is the name of the shape that the atoms in graphene are arranged in?
d) Does graphene conduct electricity? Explain your answer.
4) The diagram below shows graphite.
a) Are there strong or weak forces of attraction between the layers of graphene in graphite?
b) Do the layers easily slide over each other?
c) Does graphite conduct electricity? Explain your answer.
d) Does graphite have a very high or a very low melting and boiling point? Explain your answer.
e) Is graphite a good lubricating material? Explain your answer.
f) Is graphite a good conductor of thermal energy? Explain your answer.
5) Give 3 uses of fullerenes.
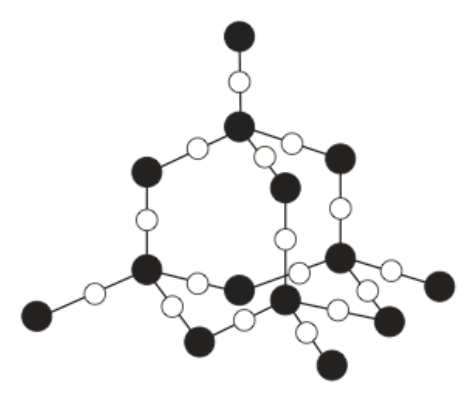
6) What is the name of the substance below?
b) Do the layers easily slide over each other?
c) Does graphite conduct electricity? Explain your answer.
d) Does graphite have a very high or a very low melting and boiling point? Explain your answer.
e) Is graphite a good lubricating material? Explain your answer.
f) Is graphite a good conductor of thermal energy? Explain your answer.
5) Give 3 uses of fullerenes.
6) What is the name of the substance below?