C2: Quiz 9 – Answers
2)
3)
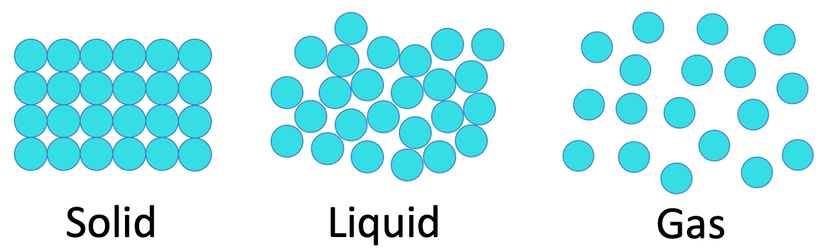
a) Solid
b) Gas
c) Liquid & gas
d) No
e) Yes
f) The particles vibrate around their fixed positions
4) The two drawbacks are:
- The particles in real life are not solid spheres; instead they are atoms, molecules and ions
- The model does not tell us anything about how strong the forces are between each of the particles; it does not explain why certain substances have weak forces (low melting & boiling points) and other substances have stronger forces (high melting & boiling points)
5)
a) Aqueous means dissolved in water
b)
Solid – (s)
Liquid – (l)
Gas – (g)
Aqueous – (aq)
1) What are the three states of matter?
2) We can use particle theory to explain the arrangement of the particles in the three different states. Draw 3 diagrams for the arrangement of the particles in the three different states.
3)
a) In which of the three states is the forces of attraction between the particles at their strongest?
b) Let’s suppose that we have H2O. In which state will the H2O particles have the most energy.
c) In which state(s) can the particles move?
d) Can liquids be compressed?
e) Can gases be compressed?
f) How would the particle theory model describe the movement of the particles in a solid?
4) Give two drawbacks of the particle model theory.
5)
a) What does aqueous mean?
b) Write down the state symbols for the following:
Solid –
Liquid –
Gas –
Aqueous –