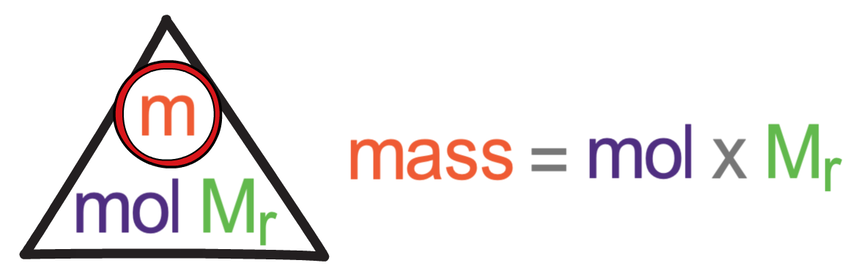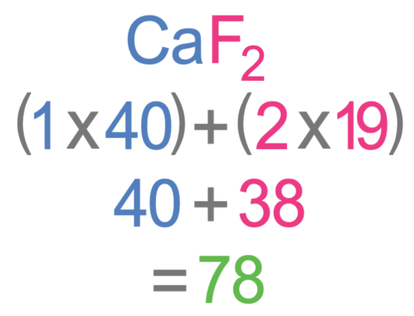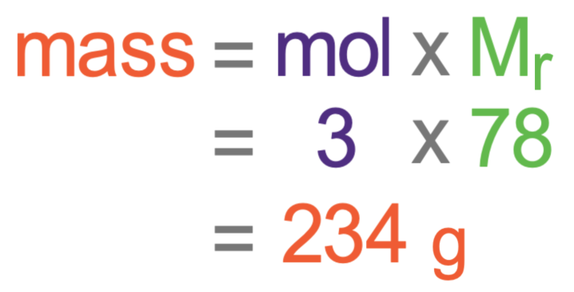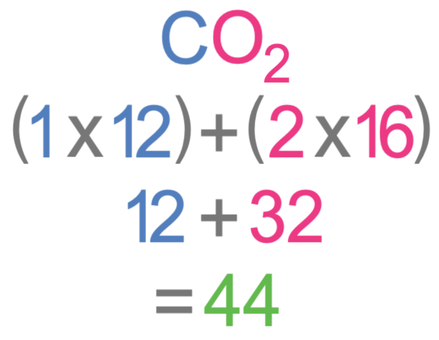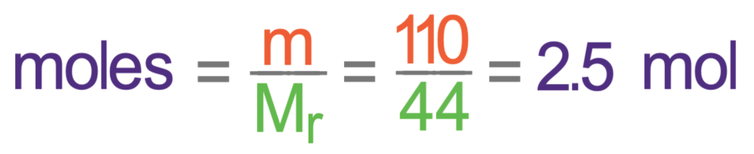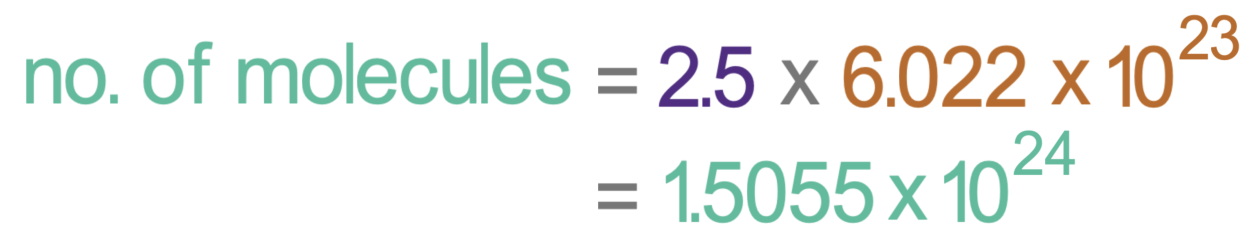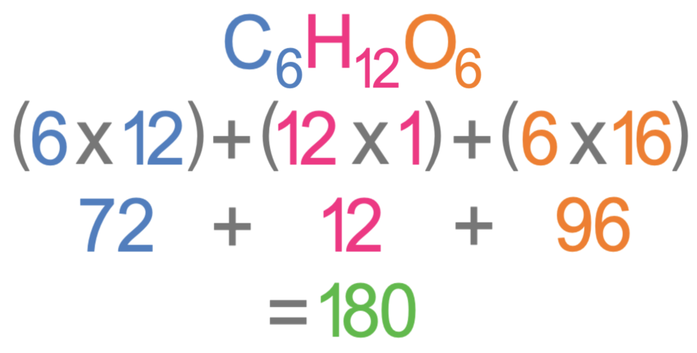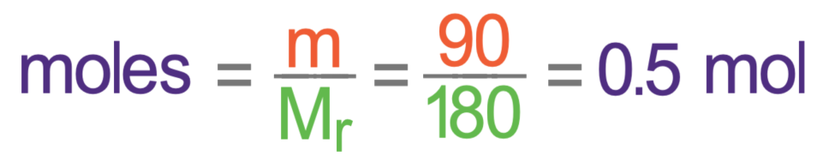C3 C) Moles
Atoms are extremely small in size and they have an extremely small mass. Scientist have created a unit of measurement known as moles to work out how many atoms there are in a certain amount of a substance. 1 mole of a substance is 602 200 000 000 000 000 000 000 atoms of that substance (or 6.022 x 1023). This number is known as Avogadro constant. Avogadro constant seems like a strange number, but it is a very useful number because it means that 1 mole of a substance is the relative formula mass (Mr) or relative atomic mass (Ar) of that substance in grams.
For example, sodium has the relative atomic mass of 23 (Ar = 23). 1 mole of a substance will be its Ar (or Mr) in grams, so 1 mole of sodium will be 23 grams. Also, 23 grams of sodium will contain 6.022 x 1023 sodium atoms.
Carbon has a relative atomic mass of 12 (Ar = 12). 1 mole of a substance will be its Ar (or Mr) in grams, so 1 mole of carbon will be 12 grams. 2 moles of carbon would be 24 grams, 3 moles of carbon would be 36 grams etc…
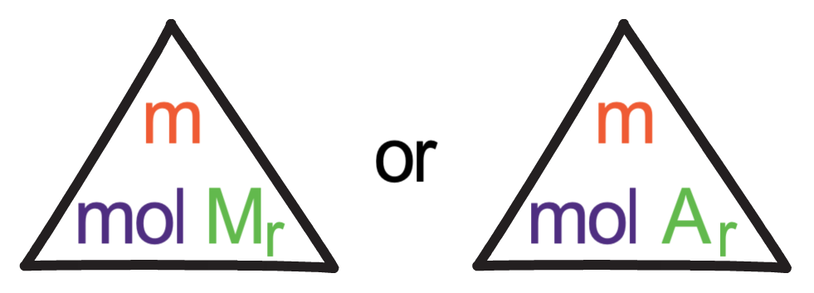
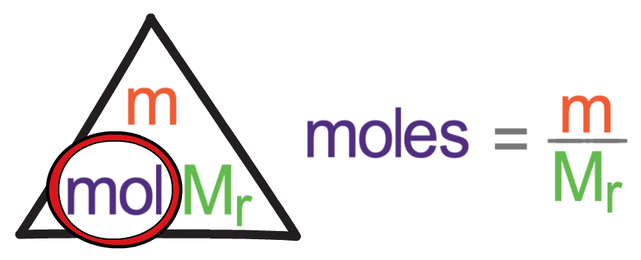
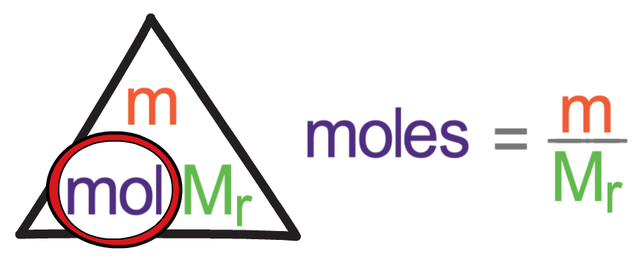
The formula triangle that relates moles, mass and Mr (or Ar) is shown below.
In the formula triangle above, Mr and Ar can be changed depending on whether we are working with a molecule or an atom. If we are working with a molecule, we use the relative formula mass (Mr), and if we are working with an atom, we use the relative atomic mass (Ar).
The above formula triangle for moles works in exactly the same way as all of the other formula triangles. We cover up what we are looking for in the formula triangle to find the calculation that we need to undertake. We then sub the values that we are given into the formula triangle.
Find the mass of 3 moles of calcium fluoride (CaF2).
The Ar of calcium (Ca) is 40 and the Ar of fluorine (F) is 19.
The first step in answering this question is to use the formula triangle to find out the calculation that we need to undertake. We are looking for mass, so we cover mass up in the formula triangle. When we do this, we see that we find the mass by multiplying moles by Mr.
The question tells us that we are looking for 3 moles. We do not know what the Mr of calcium fluoride is, but we can work it out. We work out the relative formula mass (Mr) by multiplying the number of each of the different elements by the relative atomic mass (Ar) for that element. We then add the outcomes up. In CaF2, there is one calcium atom, which has an Ar of 40, and two fluorine atoms, which have an Ar of 19. The working for finding the Mr is shown below.
The relative formula mass of calcium is 78.
We now have everything we need for the calculation for working out the mass of 3 moles of calcium fluoride; the moles are 3 and the Mr is 78.
How many moles are there in 110 grams of carbon dioxide (CO2).
The Ar of carbon (C) is 12 and the Ar of oxygen (O) is 16.
Like with the previous question, the first step is to use the formula triangle to work out the calculation that we need to undertake in order to find the number of moles. When we cover up moles in the formula triangle, it tells us that we work out the number of moles by dividing the mass by the relative formula mass (Mr).
We are told in the question that the mass of carbon dioxide is 110 grams. We do not know what the relative formula mass of carbon dioxide is, but we can work it out. We work out the relative formula mass of carbon dioxide, by multiplying the number of each of the different elements by the relative atomic mass for that element. In carbon dioxide, there is one carbon atom, which has an Ar of 12, and two oxygen atoms, which have an Ar of 16. The working for the Mr is shown below.
We now have the mass of carbon dioxide (110 grams) and the relative formula mass (44). We sub these values into the calculation to find the number of moles.
Working out the Number of Molecules
Mole questions can be extended by being asked to work out the number of molecules that are in a certain number of moles. Let’s work out the number of molecules that there are in 2.5 moles of carbon dioxide.
We know that one mole of a substance is 6.022 x 1023 number of molecules if it is a compound (or 6.022 x 1023 number of atoms if it is an element). We can work out the number of molecules of carbon dioxide there are in 2.5 moles of carbon dioxide by multiplying the number of moles that we have (2.5) by the number of molecules that there are in one mole (6.022 x 1023).
This tells us that there are 1.5055 x 1024 number of carbon dioxide molecules in 2.5 moles of carbon dioxide.
Questions where we are being asked to find the number of molecules there are in a certain number of moles are fairly straight forward; we just multiply the number of moles that we have by the number of molecules that are in 1 mole (which will always be 6.022 x 1023).
How many glucose molecules are there in 90 grams of glucose?
Glucose has the chemical formula C6H12O6. The Ar of carbon is 12, the Ar of hydrogen is 1 and the Ar of oxygen is 16.
In order to work out the number of molecules of glucose that we have in 90 grams of glucose, we must work out the number of moles of glucose that we have. We then multiply the number of moles of glucose that we have by 6.022 x 1023 (which is the number of molecules in 1 mole).
We use the moles formula triangle to work out the number of moles that we have in 90 grams of glucose. We find the calculation that we undertake by covering up moles in the formula triangle. When we cover up moles in the formula triangle, it tells us that we work out the number of moles by dividing the mass by the relative formula mass (Mr).
We are told in the question that the mass of glucose is 90 g. We do not know what the relative formula mass (Mr) of glucose is, but we can work it out. We work out the relative formula mass of glucose, by multiplying the number of each of the different elements by the relative atomic mass for that element. In glucose, there are six carbon atoms which have an Ar of 12, twelve hydrogen atoms which have an Ar of 1, and six oxygen atoms which have an Ar of 16. The working for the Mr is shown below.
The relative formula mass for glucose is 180. We now have everything we need to work out the number of moles of glucose that we have in 90 grams; we sub in 90 for the mass and 180 for the Mr.
This tells us that we have 0.5 moles of glucose.
We want to find the number of molecules of glucose that we have, and we do this by multiplying the number of moles (0.5) by the number of molecules in 1 mole (6.022 x 1023).
There are 3.011 x 1023 number of glucose molecules in 90 grams of glucose.