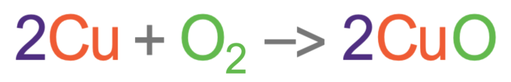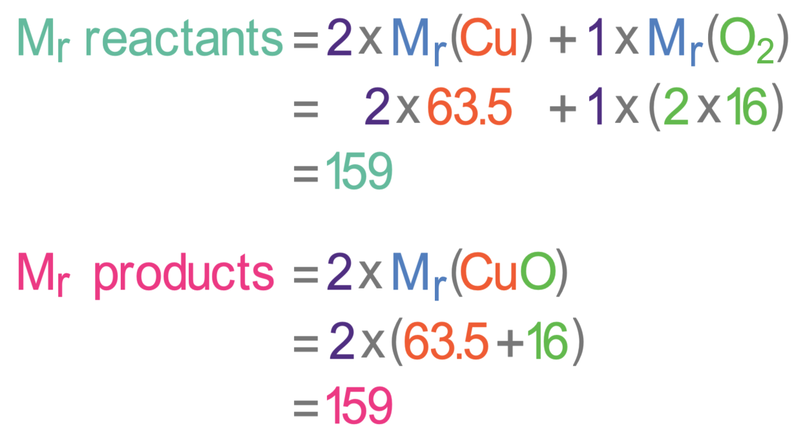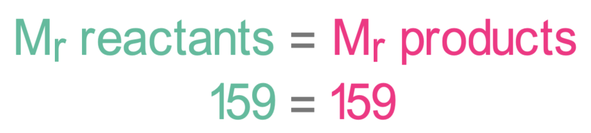Back to C3 Home
C3 E) Conservation of Mass
C3 E) Conservation of Mass
With chemical reactions, no atoms are created or destroyed. This means that the mass of the reactants will be the same as the mass of the products. Also, there will be the same number of each type of the atoms in the reactants and the products. This is known as the law of conservation of mass; this will be true for relative mass and actual mass. We are going to have a look at an example for relative mass first.
Example 1
The equation below is the reaction of copper and oxygen to create copper oxide.
The equation below is the reaction of copper and oxygen to create copper oxide.
This equation is balanced because there are 2 coppers and 2 oxygens in both the reactants (left) and the products (right). Let’s now look at the masses and see whether the law of conservation of mass holds. The relative atomic mass of copper (Cu) is 63.5 and the relative atomic mass of oxygen (O) is 16. The working for the relative mass of all of the reactants and products is shown below.
From the above working, we can see that the relative mass for all of the reactants is 159, and the relative mass for all of the products is 159. This shows us that the law of conservation of mass holds as no atoms are created or destroyed.
Mass Seems to Change
Sometimes for certain reactions in unsealed vessels, the masses of the reactants and products will appear to be different. This can be the case because either the reactants or the products may be a gas and the gas will not be part of the unsealed reaction vessel, and will therefore not be included in the mass of the reaction vessel. This will result in the mass of the reactants and products appearing to be different. There are two different scenarios.
1) Mass of reactants is less than the mass of the products
This happens when one of the reactants is a gas. This is because if the reaction vessel is not sealed, the gas that is used in the reaction will not be part of the mass of the reactants. This means that the mass of the products will appear to be greater than the mass of the reactants.
An example of a reaction where this would take place is the burning of magnesium in air (oxygen). When magnesium burns, it reacts with oxygen in the air to form magnesium oxide. The balanced equation is shown below:
Sometimes for certain reactions in unsealed vessels, the masses of the reactants and products will appear to be different. This can be the case because either the reactants or the products may be a gas and the gas will not be part of the unsealed reaction vessel, and will therefore not be included in the mass of the reaction vessel. This will result in the mass of the reactants and products appearing to be different. There are two different scenarios.
1) Mass of reactants is less than the mass of the products
This happens when one of the reactants is a gas. This is because if the reaction vessel is not sealed, the gas that is used in the reaction will not be part of the mass of the reactants. This means that the mass of the products will appear to be greater than the mass of the reactants.
An example of a reaction where this would take place is the burning of magnesium in air (oxygen). When magnesium burns, it reacts with oxygen in the air to form magnesium oxide. The balanced equation is shown below:
If the reaction is taking place in an unsealed reaction vessel, the oxygen will not be part of the reaction vessel in the reactants and therefore will not be accounted for in the mass of the reactants. After the reaction, the oxygen will become part of the reaction vessel and will therefore be include in the mass of the products. This is why the mass of the products will seem to be greater than the mass of the reactants.
2) Mass of products is less than the mass of the reactants
This happens when one of the products is a gas. This is because if the reaction vessel is not sealed, the gas that is produced is able to escape and therefore will not be part of the mass of the products. This means that the mass of the reactants will appear to be greater than the mass of the products.
An example of where this happens is the thermal decomposition of calcium carbonate. When calcium carbonate is heated up, it decomposes to become calcium oxide and carbon dioxide. The balanced equation is shown below:
2) Mass of products is less than the mass of the reactants
This happens when one of the products is a gas. This is because if the reaction vessel is not sealed, the gas that is produced is able to escape and therefore will not be part of the mass of the products. This means that the mass of the reactants will appear to be greater than the mass of the products.
An example of where this happens is the thermal decomposition of calcium carbonate. When calcium carbonate is heated up, it decomposes to become calcium oxide and carbon dioxide. The balanced equation is shown below:
The carbon dioxide produced is a gas and if the reaction takes places in an unsealed reaction vessel, the carbon dioxide will escape, which will result in the mass of the products in the reaction vessel being less than the mass of the reactants in the reaction vessel.
End Note
For the two examples above, the law of conservation of mass still holds meaning that the mass of the reactants and products are the same. It is just that the mass of the reactants and products appear to be different because some of the reactants or products are gasses and are therefore not included in the mass of the reaction vessel.
For the two examples above, the law of conservation of mass still holds meaning that the mass of the reactants and products are the same. It is just that the mass of the reactants and products appear to be different because some of the reactants or products are gasses and are therefore not included in the mass of the reaction vessel.