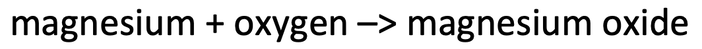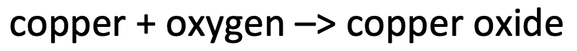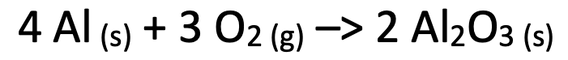Back to C3 Home
C3: Quiz 5
C3: Quiz 5
1) What is the law of conservation of mass?
2) Magnesium is burnt in air (oxygen) to produce magnesium oxide. The word equation for this reaction is shown below.
2) Magnesium is burnt in air (oxygen) to produce magnesium oxide. The word equation for this reaction is shown below.
If we reacted 24 grams of magnesium with 16 grams of oxygen, what would be the mass of magnesium oxide produced? Assume that all of the magnesium reacted with all of the oxygen.
3) The word equation below shows the reaction of copper with oxygen to produce copper oxide.
3) The word equation below shows the reaction of copper with oxygen to produce copper oxide.
I reacted 31.75 grams of copper and obtained 39.75 grams of copper oxide. Find the mass of oxygen that reacted. Assume that all of the copper reacted.
4) A student heated some calcium carbonate in an open crucible. The equation for the reaction is shown below.
4) A student heated some calcium carbonate in an open crucible. The equation for the reaction is shown below.
The student measured the mass of the crucible before and after the reaction. Would the mass of the crucible increase or decrease during the reaction? Explain your answer.
5) When aluminium powder is heated, it reacts with oxygen to form aluminium oxide powder. The equation for this reaction is shown below.
5) When aluminium powder is heated, it reacts with oxygen to form aluminium oxide powder. The equation for this reaction is shown below.
A student carried out this experiment. He measured the mass of the powder before and after the reaction. Would the mass of the powder be greater before or after the reaction? Explain your answer.
6) For the reactions in questions 4 and 5, does the law of conservation of mass still work? Explain your answer.
6) For the reactions in questions 4 and 5, does the law of conservation of mass still work? Explain your answer.