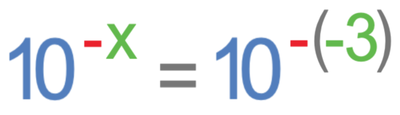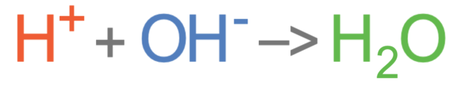C4 A) Acids & Alkalis
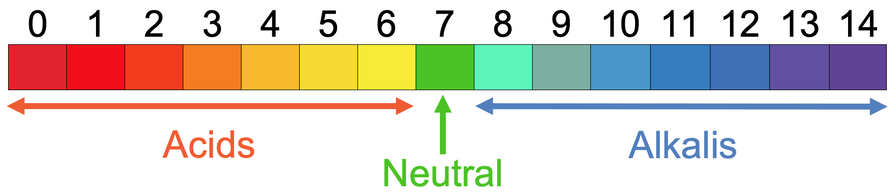
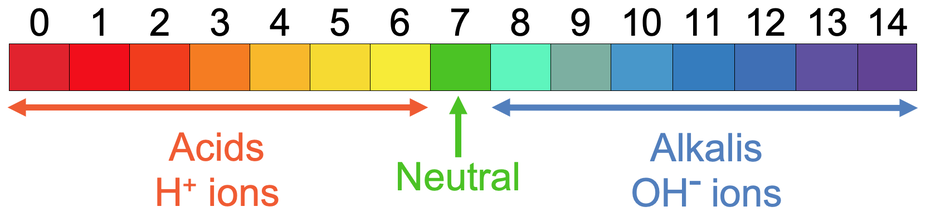
- 7 on the pH scale is neutral.
- 0 to 6 are acids. The lower the pH value is, the more acidic the solution is – 0 is the most acidic.
- 8 to 14 are alkaline. The higher the pH value is, the more alkaline the solution is – 14 is the most alkaline solution.
Measuring the pH of Solutions
We are able to find the pH of a solution by using an indicator, such as universal indicator (universal indicator can be a solution or a piece of paper). We find the pH of a solution with universal indicator as a solution by adding a few drops of universal indicator to the solution that we are testing. We then wait to see what colour the solution with the universal indicator in turns. Universal indicator will be a range of colours depending on the pH of the solution; it will be green for neutral solutions, orange/ red for acidic solutions, and blue/ purple for alkaline solutions. The full range of colours for universal indicator are shown on the above pH number line. When we are using universal indicator to see what pH a solution is, we will have a colour chart with the different pHs values on. We then place the colour chart next to our solution with universal indicator in to see what pH the solution is.
There are many other indicators in addition to universal indicator that we can use to find the pH of a solution.
We can also measure the pH of a solution by using an electronic pH probe. We do this by placing the probe into the solution and looking at the digital screen to see the pH value. Working out the pH of a solution with a pH probe is more accurate than using an indicator.
The Ions for Acids
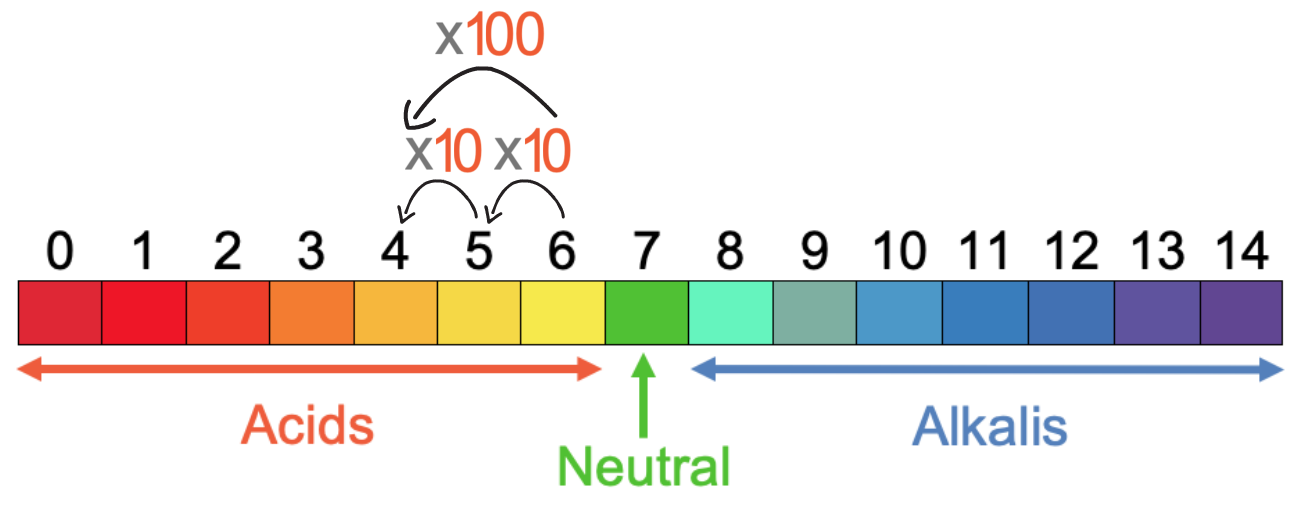
An acid is a substance that forms H+ ions when dissolved in water; H+ ions are known as positive hydrogen ions (they are essentially a proton). A more acidic solution will have a greater concentration of H+ ions. As we move down the pH values by 1 for acids, the concentration of H+ ions increases by a factor of 10. So, the concentration of H+ ions is 10 times greater in a solution with a pH of 5 compared to a solution with a pH of 6. And, the concentration of H+ ions is 10 times greater in a solution with a pH of 4 compared to a solution with a pH of 5. This means that the concentration of H+ ions is 100 times greater in a solution with a pH of 4 compared to a solution with a pH of 6; to go from a pH of 6 to a pH of 4, we multiply the concentration of H+ ions by 10 two times, which means that we multiply by 100.
There is a general rule for this, but it is quite confusing so you may prefer to just remember that as the pH goes down by 1 for acids, the concentration of H+ ions increases by a factor of 10. The rule is shown below:
x in the above formula is the change in pH for the acid.
Example 1
By what factor does the H+ ion concentration change by when a solution goes from a pH of 5 to a pH of 2?
We find the factor that the H+ ion concentration changes by when a solution goes from a pH of 5 to a pH of 2 by using the formula below.
x in the above formula is the change in pH. The pH is changing from 5 to 2, which means that the pH decreases by 3; the value of x is -3. Therefore, we sub -3 as x into the formula.
A double negative makes a positive, which means that –(-3) becomes 3.
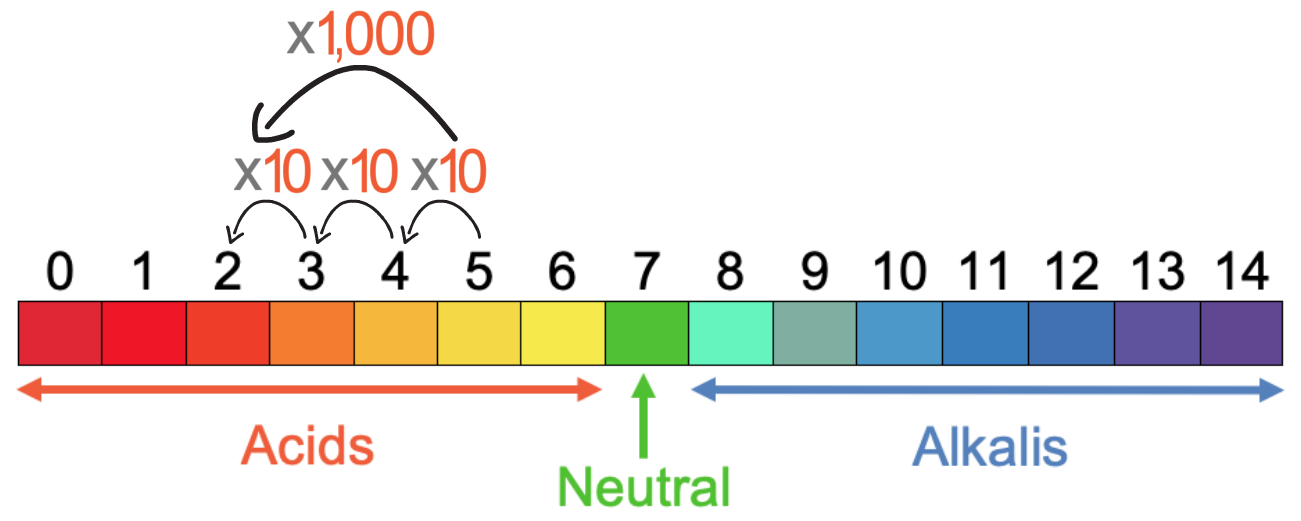
This tells us that the concentration of H+ ions increases by a factor of 1,000 when the solution goes from a pH of 5 to a pH of 2; the concentration of H+ ions is 1,000 times greater when the solution has a pH of 2 compared to when the solution had a pH of 5.
From undertaking this process, you can see that using the formula to work out the factor that the concentration of H+ ions changes by is a little bit tricky. Therefore, you may prefer to work it out by remembering the rule, which is that every time the pH goes down by 1, the concentration of H+ ions increases by a factor of 10. The working for the pH going from 5 to 2 is shown below.
This tells us that the concentration of H+ ions increases by a factor of 1,000 when the solution goes from a pH of 5 to a pH of 2.
Bases
A base is a substance with a pH that is greater than 7. Alkalis are bases that dissolve in water. When alkalis dissolve in water, they produce OH- ions, which are known as negative hydroxide ions.
So, acids contain H+ ions (positive hydrogen ions) and alkalis contain OH- ions (negative hydroxide ions). I have added these ions to the pH number line below.
Neutralisation Reactions
Acids contain H+ ions (positive hydrogen ions) and bases/ alkalis contain OH- ions (negative hydroxide ions). A neutralisation reaction is where an acid reacts with a base to produce salt and water. The word equation is shown below:
We can also write the reaction out in terms of the ions involved. We know that acids have H+ ions and bases have OH- ions. When an acid and base react, these ions will combine to give us water.
The products of a neutralisation reaction are neutral – the products will have a pH of 7. When we undertake this reaction, we can use an indicator to show us when the reaction is complete; if we used universal indicator, the indicator would turn green when the reaction is complete.
Bases & Alkali
A base is any substance that reacts with an acid to produce a salt and water only. Bases neutralise acids. A base can be a metal oxide or a metal hydroxide.
Bases that are soluble in water are also known as alkalis (soluble in water means that it can dissolve in water). For example, sodium hydroxide dissolves in water and reacts with acids to produce a salt and water only; this means that sodium hydroxide is an alkali.
A base that is insoluble in water is just a base and not an alkali. For example, copper oxide reacts with acids to produce a salt and water only, but it does not dissolve in water; this means that copper oxide is just a base and not an alkali.