Back to C4 Home
C4 E) Metals Reacting with Water
C4 E) Metals Reacting with Water
Most metals react with water, but the rate of reaction with water varies depending on how reactive the metal is. The standard reaction of a metal with water is:
Metal hydroxides are bases. Some metal hydroxides dissolved in water and are therefore alkalis. Metal hydroxides have a positive metal ion and a negative hydroxide ion. A negative hydroxide ion has a charge of 1- (OH-). The charge for the positive metal ion depends on the metal that is involved.
Metals that are in group 1 of the periodic table react very aggressively with water. Examples of these group 1 metals are lithium, sodium, potassium etc. Group 1 metals react so aggressively with water because they want to lose the single electron in their outermost electron shell. We can conduct an experiment of group 1 metals reacting with water by having a container filled with water and dropping the group 1 metals into it. The group 1 metals will react with the water to produce a metal hydroxide and hydrogen gas. As we go down the group 1 column, the metals become more reactive, which means that they will react more aggressively/ faster and will produce hydrogen quicker (the reasons why group 1 metals/ alkali metals become more reactive as we go down the column are explained in the group 1 section; click here to be taken to that section).
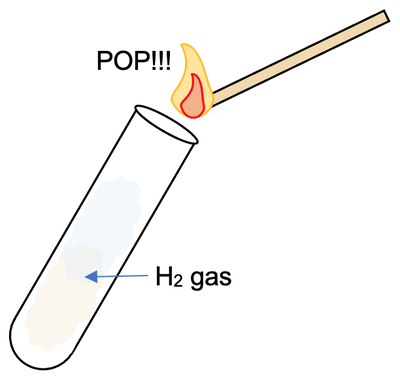
We can test for the presence of hydrogen gas by collecting some of the gas in a test tube. We then hold a lit splint above the test tube that we collected the gas in; if we hear a squeaky pop, it tells us that the gas is hydrogen (we could also hold our lit splint above where the hydrogen gas was produced rather than collecting it in a test tube).