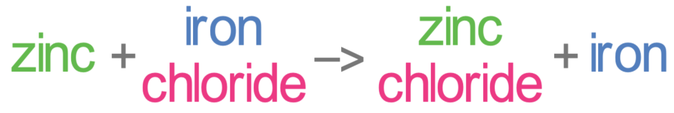C4 H) Redox Reactions
The content in this section is quite tricky, so you may need to work through it a few times.
In this section, we are going to look at redox reactions with respect to what happens to the electrons. We can use OIL RIG to help us remember what oxidation and reduction means.
Oxidation is loss of electrons and reduction is gain of electrons. In redox reactions, both REDuction and OXidation happen at the same time and this is why the reactions are called REDOX reactions.
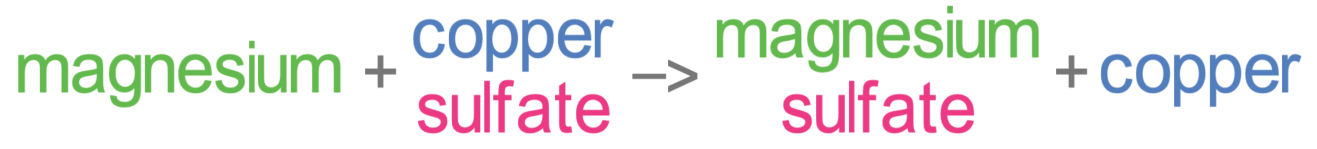
In the previous section, we looked at the displacement reaction between magnesium and copper sulfate. The word and chemical equations for this reaction are shown below.
We can now write this equation out in terms of the ions that are involved. This equation is shown below.
The sulfate ion (SO42-) is the same on both sides of the reaction arrow – this means that the sulfate ion does not take part in the reaction. Therefore, we can remove the sulfate ion from the ion equation, which results in the ion equation becoming:
We can now split this ion equation into two half equations for the two different metals.
Magnesium
Magnesium goes from neutrally charged (Mg) to positively charged by 2 (Mg2+). It does this by losing 2 electrons. The half equation for this is shown below.
Magnesium has lost electrons, which means that it has been oxidised (oxidation is loss).
Copper
Copper goes from being positively charged by 2 (Cu2+) to neutrally charged (Cu). It does this by gaining 2 electrons. The half equation for this is shown below.
Copper has gained electrons, which means that it has been reduced (reduction is gain).
The Outcomes
For displacement reactions, the metal that is doing the displacing loses electrons, which means that it is oxidised (oxidation is loss) – this was magnesium in the above reaction.
The metal that is displaced gains electrons, which means that it is reduced (reduction is gain) – this was copper in the above reaction.
Another Displacement Reaction
We are now going to have a look at the reaction of zinc with iron chloride. Zinc is a more reactive metal than iron. This means that zinc will displace the iron in iron chloride, which results in the products of the reaction being zinc chloride and iron. The word and chemical equations for this are shown below.
We can now write this equation out in terms of the ions that are involved. This equation is shown below.
The chloride ions (2 Cl-) are the same on both sides of the reaction arrow – this means that they do not take part in the reaction. Therefore, we can remove them from the ion equation, which results in the ion equation becoming:
We can now split this ion equation into two half equations.
Zinc
Zinc goes from neutrally charged (Zn) to positively charged by 2 (Zn2+). It does this by losing 2 electrons. The half equation for this is shown below.
Zinc has lost electrons, which means that it has been oxidised (oxidation is loss). Also, it is always the displacing metal that is oxidised – zinc here is the displacing metal, so zinc is oxidised.
Iron
Iron goes from being positively charged by 2 (Fe2+) to neutrally charged (Fe). It does this by gaining 2 electrons. The half equation for this is shown below.
Iron has gained electrons, which means that it has been reduced (reduction is gain). Also, it is always the displaced metal that is reduced – iron here is the displaced metal, so iron is reduced.
Outcome
So, for the reaction between zinc and iron chloride, zinc has been oxidised as it loses electrons, and iron has been reduced as it gains electrons.